Input interpretation

top 10 chemicals | by boiling point
Result
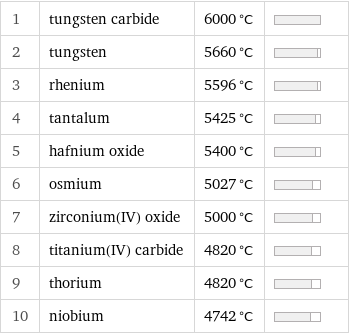
1 | tungsten carbide | 6000 °C | 2 | tungsten | 5660 °C | 3 | rhenium | 5596 °C | 4 | tantalum | 5425 °C | 5 | hafnium oxide | 5400 °C | 6 | osmium | 5027 °C | 7 | zirconium(IV) oxide | 5000 °C | 8 | titanium(IV) carbide | 4820 °C | 9 | thorium | 4820 °C | 10 | niobium | 4742 °C |
Structure diagrams
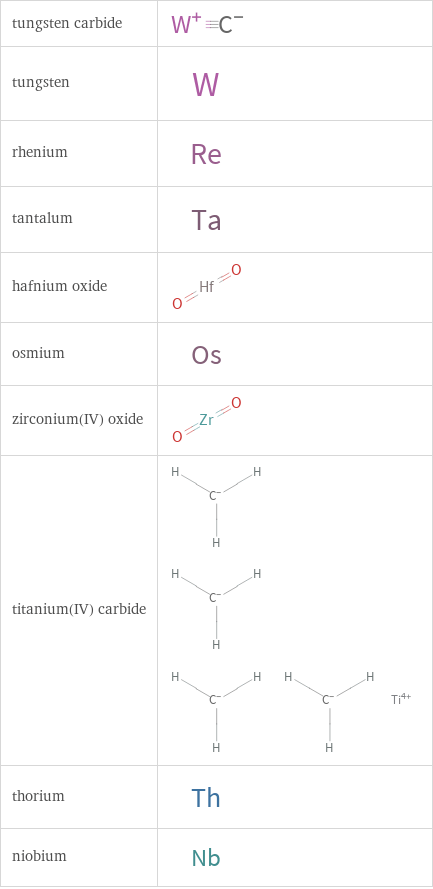
Structure diagrams
Basic properties
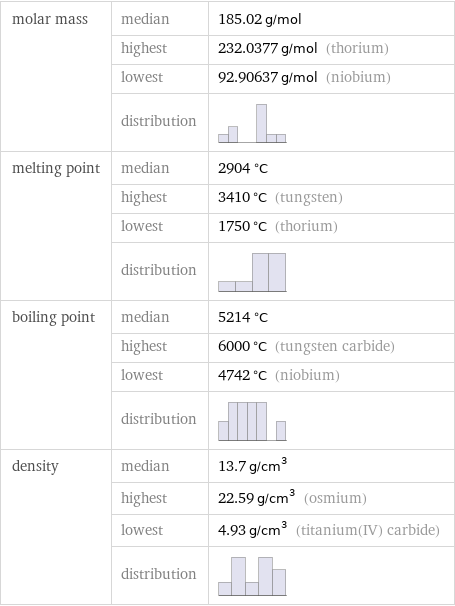
molar mass | median | 185.02 g/mol | highest | 232.0377 g/mol (thorium) | lowest | 92.90637 g/mol (niobium) | distribution | melting point | median | 2904 °C | highest | 3410 °C (tungsten) | lowest | 1750 °C (thorium) | distribution | boiling point | median | 5214 °C | highest | 6000 °C (tungsten carbide) | lowest | 4742 °C (niobium) | distribution | density | median | 13.7 g/cm^3 | highest | 22.59 g/cm^3 (osmium) | lowest | 4.93 g/cm^3 (titanium(IV) carbide) | distribution |
Units
