Input interpretation
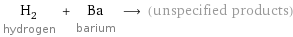
H_2 hydrogen + Ba barium ⟶ (unspecified products)
Sample reactions

H_2 hydrogen + Ba barium ⟶ BaH_2 barium hydride
Chemical names and formulas

| hydrogen | barium formula | H_2 | Ba name | hydrogen | barium IUPAC name | molecular hydrogen | barium
Substance properties
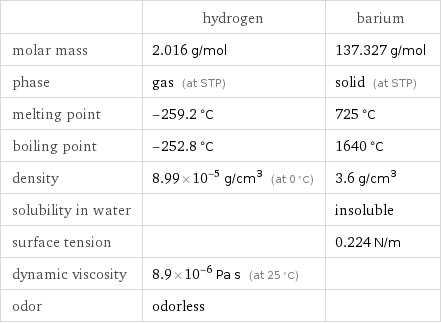
| hydrogen | barium molar mass | 2.016 g/mol | 137.327 g/mol phase | gas (at STP) | solid (at STP) melting point | -259.2 °C | 725 °C boiling point | -252.8 °C | 1640 °C density | 8.99×10^-5 g/cm^3 (at 0 °C) | 3.6 g/cm^3 solubility in water | | insoluble surface tension | | 0.224 N/m dynamic viscosity | 8.9×10^-6 Pa s (at 25 °C) | odor | odorless |
Units
