Input interpretation
strontium hydroxide | zirconium
Chemical names and formulas
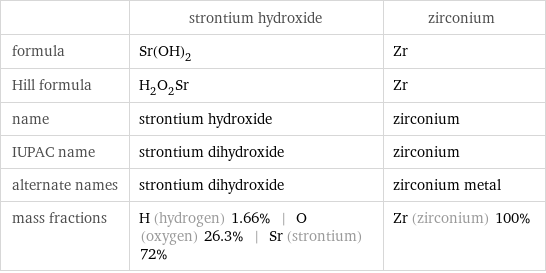
| strontium hydroxide | zirconium formula | Sr(OH)_2 | Zr Hill formula | H_2O_2Sr | Zr name | strontium hydroxide | zirconium IUPAC name | strontium dihydroxide | zirconium alternate names | strontium dihydroxide | zirconium metal mass fractions | H (hydrogen) 1.66% | O (oxygen) 26.3% | Sr (strontium) 72% | Zr (zirconium) 100%
Structure diagrams
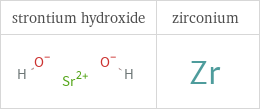
Structure diagrams
Basic properties
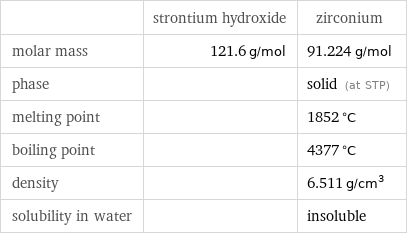
| strontium hydroxide | zirconium molar mass | 121.6 g/mol | 91.224 g/mol phase | | solid (at STP) melting point | | 1852 °C boiling point | | 4377 °C density | | 6.511 g/cm^3 solubility in water | | insoluble
Units

Thermodynamic properties
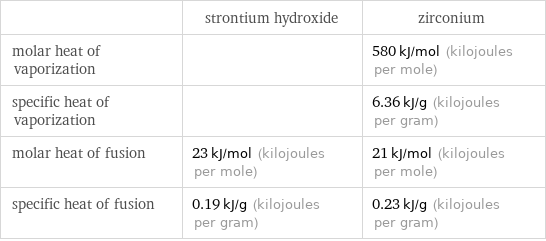
| strontium hydroxide | zirconium molar heat of vaporization | | 580 kJ/mol (kilojoules per mole) specific heat of vaporization | | 6.36 kJ/g (kilojoules per gram) molar heat of fusion | 23 kJ/mol (kilojoules per mole) | 21 kJ/mol (kilojoules per mole) specific heat of fusion | 0.19 kJ/g (kilojoules per gram) | 0.23 kJ/g (kilojoules per gram)