Input interpretation
silver chloride | vanadium(II) bromide
Chemical names and formulas
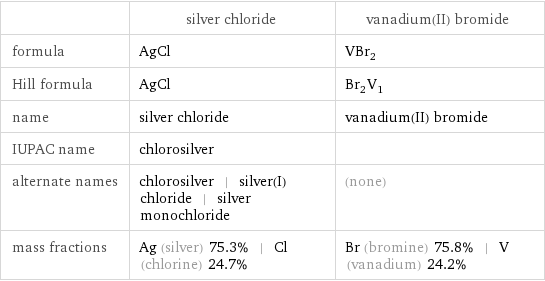
| silver chloride | vanadium(II) bromide formula | AgCl | VBr_2 Hill formula | AgCl | Br_2V_1 name | silver chloride | vanadium(II) bromide IUPAC name | chlorosilver | alternate names | chlorosilver | silver(I) chloride | silver monochloride | (none) mass fractions | Ag (silver) 75.3% | Cl (chlorine) 24.7% | Br (bromine) 75.8% | V (vanadium) 24.2%
Structure diagrams
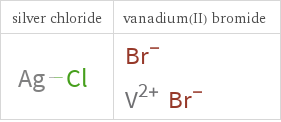
Structure diagrams
Basic properties
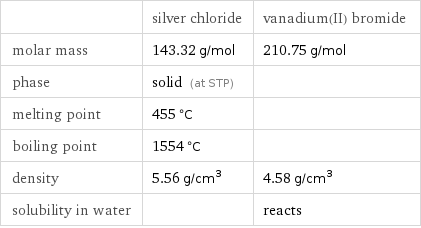
| silver chloride | vanadium(II) bromide molar mass | 143.32 g/mol | 210.75 g/mol phase | solid (at STP) | melting point | 455 °C | boiling point | 1554 °C | density | 5.56 g/cm^3 | 4.58 g/cm^3 solubility in water | | reacts
Units
