Input interpretation
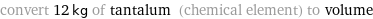
convert 12 kg of tantalum (chemical element) to volume
Results

720.72 mL (milliliters)
Possible intermediate steps

Convert from kilograms into milliliters: 12 kg of Ta (tantalum) Convert from kilograms to cubic centimeters using the relation 1000 g (grams) = 1 kg (kilogram) and the density 16.65 g/cm^3 (grams per cubic centimeter) at STP of Ta: (12 kg)/1 × (1000 g)/(1 kg) × (1 cm^3)/(16.65 g) = 720.72 cm^3 Convert to milliliters using the relation 1 cm^3 (cubic centimeter) = 1 mL (milliliter): Answer: | | (720.72 cm^3)/1 × (1 mL)/(1 cm^3) = 720.72 mL
Unit conversions
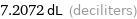
7.2072 dL (deciliters)
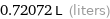
0.72072 L (liters)

720.72 cm^3 (cubic centimeters)
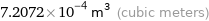
7.2072×10^-4 m^3 (cubic meters)
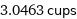
3.0463 cups
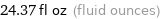
24.37 fl oz (fluid ounces)
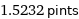
1.5232 pints
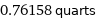
0.76158 quarts
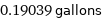
0.19039 gallons
Comparisons as volume
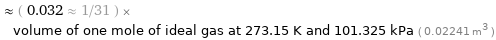
≈ ( 0.032 ≈ 1/31 ) × volume of one mole of ideal gas at 273.15 K and 101.325 kPa ( 0.02241 m^3 )
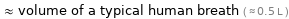
≈ volume of a typical human breath ( ≈ 0.5 L )
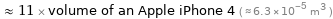
≈ 11 × volume of an Apple iPhone 4 ( ≈ 6.3×10^-5 m^3 )
Comparisons as volume of food or beverage
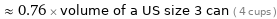
≈ 0.76 × volume of a US size 3 can ( 4 cups )
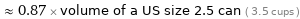
≈ 0.87 × volume of a US size 2.5 can ( 3.5 cups )
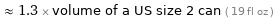
≈ 1.3 × volume of a US size 2 can ( 19 fl oz )
Interpretations
volume
volume of food or beverage
capacity
section modulus
Basic unit dimensions
![[length]^3](../image_source/a8f7e694e0724fe8dd5021b1eac1f4f5.png)
[length]^3
Corresponding quantities

Radius r of a sphere from V = 4πr^3/3: | 56 mm (millimeters) | 0.18 feet | 2.2 inches
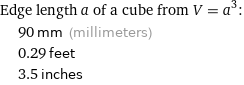
Edge length a of a cube from V = a^3: | 90 mm (millimeters) | 0.29 feet | 3.5 inches
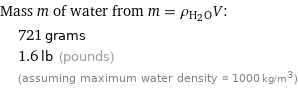
Mass m of water from m = ρ_(H_2O)V: | 721 grams | 1.6 lb (pounds) | (assuming maximum water density ≈ 1000 kg/m^3)
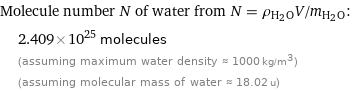
Molecule number N of water from N = ρ_(H_2O)V/m_(H_2O): | 2.409×10^25 molecules | (assuming maximum water density ≈ 1000 kg/m^3) | (assuming molecular mass of water ≈ 18.02 u)