Input interpretation
heavy water
Chemical names and formulas

formula | D_2O name | heavy water alternate names | deuterium oxide | dideuterium oxide | heavy water-d2 | water-d2 mass fractions | O (oxygen) 0.799% | H (hydrogen) 0.201%
Lewis structure

Draw the Lewis structure of heavy water. Start by drawing the overall structure of the molecule: Count the total valence electrons of the hydrogen (n_H, val = 1) and oxygen (n_O, val = 6) atoms: 2 n_H, val + n_O, val = 8 Calculate the number of electrons needed to completely fill the valence shells for hydrogen (n_H, full = 2) and oxygen (n_O, full = 8): 2 n_H, full + n_O, full = 12 Subtracting these two numbers shows that 12 - 8 = 4 bonding electrons are needed. Each bond has two electrons, so the above diagram has all the necessary bonds. There are 2 bonds and hence 4 bonding electrons in the diagram. Lastly, fill in the remaining unbonded electrons on each atom. In total, there remain 8 - 4 = 4 electrons left to draw: Answer: | |
3D structure

3D structure
Basic properties
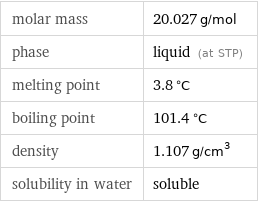
molar mass | 20.027 g/mol phase | liquid (at STP) melting point | 3.8 °C boiling point | 101.4 °C density | 1.107 g/cm^3 solubility in water | soluble
Units

Liquid properties (at STP)
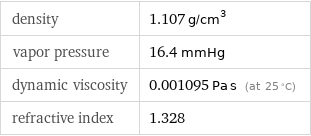
density | 1.107 g/cm^3 vapor pressure | 16.4 mmHg dynamic viscosity | 0.001095 Pa s (at 25 °C) refractive index | 1.328
Units
Thermodynamic properties

molar heat of fusion | 6.2 kJ/mol specific heat of fusion | 0.3096 kJ/g (at STP)
Non-standard atom properties

H-2 | 2
Chemical identifiers
CAS number | 7789-20-0 PubChem CID number | 24602 PubChem SID number | 24849176 SMILES identifier | O InChI identifier | InChI=1/H2O/h1H2/i/hD2/f/i1D2 RTECS number | ZC0230000 MDL number | MFCD00044636
NFPA label

NFPA label

NFPA health rating | 0 NFPA fire rating | 0 NFPA reactivity rating | 0
Toxicity properties

odor | odorless
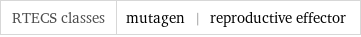
RTECS classes | mutagen | reproductive effector