Input interpretation
calcium hydroxide
Chemical names and formulas

formula | Ca(OH)_2 Hill formula | CaH_2O_2 name | calcium hydroxide IUPAC name | calcium dihydroxide alternate names | calcium dihydroxide | calcium hydrate | hydrated lime | slaked lime mass fractions | Ca (calcium) 54.1% | H (hydrogen) 2.72% | O (oxygen) 43.2%
Structure diagram
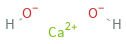
Structure diagram
Basic properties
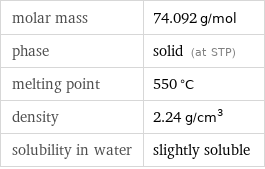
molar mass | 74.092 g/mol phase | solid (at STP) melting point | 550 °C density | 2.24 g/cm^3 solubility in water | slightly soluble
Units

Solid properties (at STP)

density | 2.24 g/cm^3 refractive index | 1.555
Units

Thermodynamic properties

specific heat capacity c_p | solid | 1.181 J/(g K) molar heat capacity c_p | solid | 87.5 J/(mol K) specific free energy of formation Δ_fG° | solid | -12.11 kJ/g molar free energy of formation Δ_fG° | solid | -897.5 kJ/mol specific heat of formation Δ_fH° | solid | -13.3 kJ/g molar heat of formation Δ_fH° | solid | -985.2 kJ/mol specific entropy S° | solid | 1.12 J/(g K) molar entropy S° | solid | 83 J/(mol K) (at STP)
Chemical identifiers
![CAS number | 1305-62-0 PubChem CID number | 14777 PubChem SID number | 24868536 SMILES identifier | [OH-].[OH-].[Ca+2] InChI identifier | InChI=1/Ca.2H2O/h;2*1H2/q+2;;/p-2/fCa.2HO/h;2*1h/qm;2*-1 RTECS number | EW2800000 MDL number | MFCD00010901](../image_source/0a9c0b3184eba609b69c259ac5db2b2b.png)
CAS number | 1305-62-0 PubChem CID number | 14777 PubChem SID number | 24868536 SMILES identifier | [OH-].[OH-].[Ca+2] InChI identifier | InChI=1/Ca.2H2O/h;2*1H2/q+2;;/p-2/fCa.2HO/h;2*1h/qm;2*-1 RTECS number | EW2800000 MDL number | MFCD00010901
NFPA label

NFPA label

NFPA health rating | 3 NFPA fire rating | 0 NFPA reactivity rating | 0
Toxicity properties

odor | odorless lethal dosage | 7340 mg/kg (oral dose for rats)

probable lethal dose for man | 1 L (liter) long-term exposure limit | 5 mg/m^3 (over 8 hours) RTECS classes | agricultural chemical and pesticide | mutagen | primary irritant
Ion equivalents

Ca^(2+) (calcium cation) | 1 (OH)^- (hydroxide anion) | 2