Input interpretation
vanadium(IV) oxide sulfate hydrate | potassium alum
Chemical names and formulas
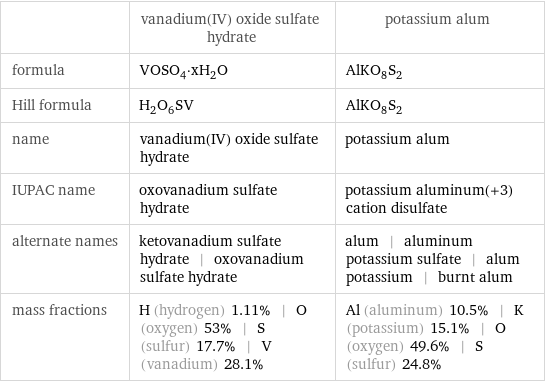
| vanadium(IV) oxide sulfate hydrate | potassium alum formula | VOSO_4·xH_2O | AlKO_8S_2 Hill formula | H_2O_6SV | AlKO_8S_2 name | vanadium(IV) oxide sulfate hydrate | potassium alum IUPAC name | oxovanadium sulfate hydrate | potassium aluminum(+3) cation disulfate alternate names | ketovanadium sulfate hydrate | oxovanadium sulfate hydrate | alum | aluminum potassium sulfate | alum potassium | burnt alum mass fractions | H (hydrogen) 1.11% | O (oxygen) 53% | S (sulfur) 17.7% | V (vanadium) 28.1% | Al (aluminum) 10.5% | K (potassium) 15.1% | O (oxygen) 49.6% | S (sulfur) 24.8%
Structure diagrams
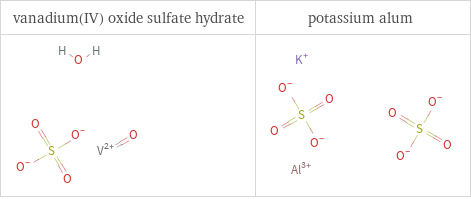
Structure diagrams
Basic properties

| vanadium(IV) oxide sulfate hydrate | potassium alum molar mass | 181.01 g/mol | 258.19 g/mol phase | solid (at STP) | solid (at STP) melting point | 105 °C | 92.5 °C density | | 1.725 g/cm^3
Units
