Input interpretation
zinc carbonate
Chemical names and formulas

formula | ZnCO_3 Hill formula | CO_3Zn name | zinc carbonate alternate names | natural smithsonite | zinc carbonate(smithsonite) mass fractions | C (carbon) 9.58% | O (oxygen) 38.3% | Zn (zinc) 52.1%
Structure diagram
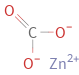
Structure diagram
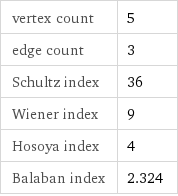
vertex count | 5 edge count | 3 Schultz index | 36 Wiener index | 9 Hosoya index | 4 Balaban index | 2.324
Basic properties
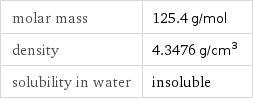
molar mass | 125.4 g/mol density | 4.3476 g/cm^3 solubility in water | insoluble
Units

Thermodynamic properties
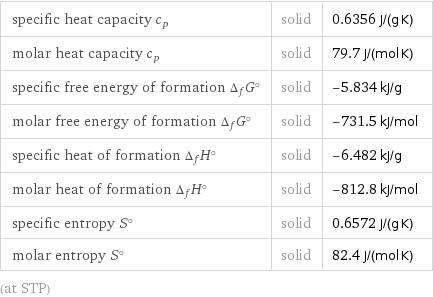
specific heat capacity c_p | solid | 0.6356 J/(g K) molar heat capacity c_p | solid | 79.7 J/(mol K) specific free energy of formation Δ_fG° | solid | -5.834 kJ/g molar free energy of formation Δ_fG° | solid | -731.5 kJ/mol specific heat of formation Δ_fH° | solid | -6.482 kJ/g molar heat of formation Δ_fH° | solid | -812.8 kJ/mol specific entropy S° | solid | 0.6572 J/(g K) molar entropy S° | solid | 82.4 J/(mol K) (at STP)
Chemical identifiers
![CAS number | 3486-35-9 PubChem CID number | 19005 SMILES identifier | C(=O)([O-])[O-].[Zn+2] InChI identifier | InChI=1/CH2O3.Zn/c2-1(3)4;/h(H2, 2, 3, 4);/q;+2/p-2/fCO3.Zn/q-2;m EU number | 222-477-6 Gmelin number | 233424 RTECS number | FG3375000](../image_source/b5713879762e9f3da5873fb9b23536d2.png)
CAS number | 3486-35-9 PubChem CID number | 19005 SMILES identifier | C(=O)([O-])[O-].[Zn+2] InChI identifier | InChI=1/CH2O3.Zn/c2-1(3)4;/h(H2, 2, 3, 4);/q;+2/p-2/fCO3.Zn/q-2;m EU number | 222-477-6 Gmelin number | 233424 RTECS number | FG3375000
NFPA label

NFPA label

NFPA health rating | 1 NFPA fire rating | 0 NFPA reactivity rating | 0