Input interpretation

cerium (chemical element) | erbium (chemical element) | holmium (chemical element)
Periodic table location
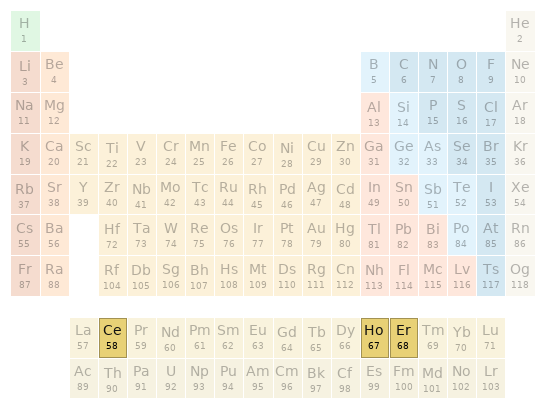
Periodic table location
Images

Images
Basic elemental properties
![| cerium | erbium | holmium atomic symbol | Ce | Er | Ho atomic number | 58 | 68 | 67 short electronic configuration | [Xe]6s^24f^15d^1 | [Xe]6s^24f^12 | [Xe]6s^24f^11 Aufbau diagram | 5d 4f 6s | 4f 6s | 4f 6s block | f | f | f period | 6 | 6 | 6 atomic mass | 140.116 u | 167.259 u | 164.93033 u half-life | (stable) | (stable) | (stable)](../image_source/8c8f1391ad235a0ba61117e836e394b9.png)
| cerium | erbium | holmium atomic symbol | Ce | Er | Ho atomic number | 58 | 68 | 67 short electronic configuration | [Xe]6s^24f^15d^1 | [Xe]6s^24f^12 | [Xe]6s^24f^11 Aufbau diagram | 5d 4f 6s | 4f 6s | 4f 6s block | f | f | f period | 6 | 6 | 6 atomic mass | 140.116 u | 167.259 u | 164.93033 u half-life | (stable) | (stable) | (stable)
Thermodynamic properties
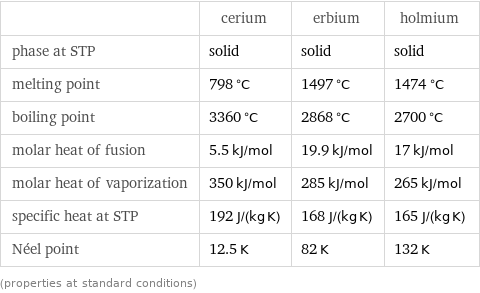
| cerium | erbium | holmium phase at STP | solid | solid | solid melting point | 798 °C | 1497 °C | 1474 °C boiling point | 3360 °C | 2868 °C | 2700 °C molar heat of fusion | 5.5 kJ/mol | 19.9 kJ/mol | 17 kJ/mol molar heat of vaporization | 350 kJ/mol | 285 kJ/mol | 265 kJ/mol specific heat at STP | 192 J/(kg K) | 168 J/(kg K) | 165 J/(kg K) Néel point | 12.5 K | 82 K | 132 K (properties at standard conditions)
Material properties
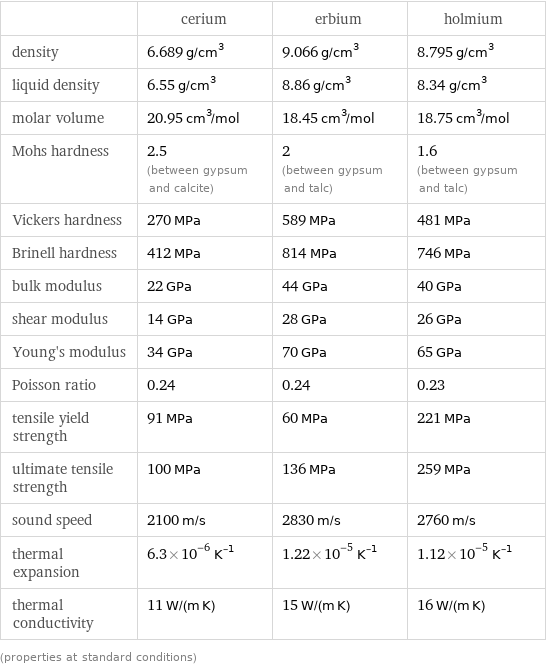
| cerium | erbium | holmium density | 6.689 g/cm^3 | 9.066 g/cm^3 | 8.795 g/cm^3 liquid density | 6.55 g/cm^3 | 8.86 g/cm^3 | 8.34 g/cm^3 molar volume | 20.95 cm^3/mol | 18.45 cm^3/mol | 18.75 cm^3/mol Mohs hardness | 2.5 (between gypsum and calcite) | 2 (between gypsum and talc) | 1.6 (between gypsum and talc) Vickers hardness | 270 MPa | 589 MPa | 481 MPa Brinell hardness | 412 MPa | 814 MPa | 746 MPa bulk modulus | 22 GPa | 44 GPa | 40 GPa shear modulus | 14 GPa | 28 GPa | 26 GPa Young's modulus | 34 GPa | 70 GPa | 65 GPa Poisson ratio | 0.24 | 0.24 | 0.23 tensile yield strength | 91 MPa | 60 MPa | 221 MPa ultimate tensile strength | 100 MPa | 136 MPa | 259 MPa sound speed | 2100 m/s | 2830 m/s | 2760 m/s thermal expansion | 6.3×10^-6 K^(-1) | 1.22×10^-5 K^(-1) | 1.12×10^-5 K^(-1) thermal conductivity | 11 W/(m K) | 15 W/(m K) | 16 W/(m K) (properties at standard conditions)
Electromagnetic properties
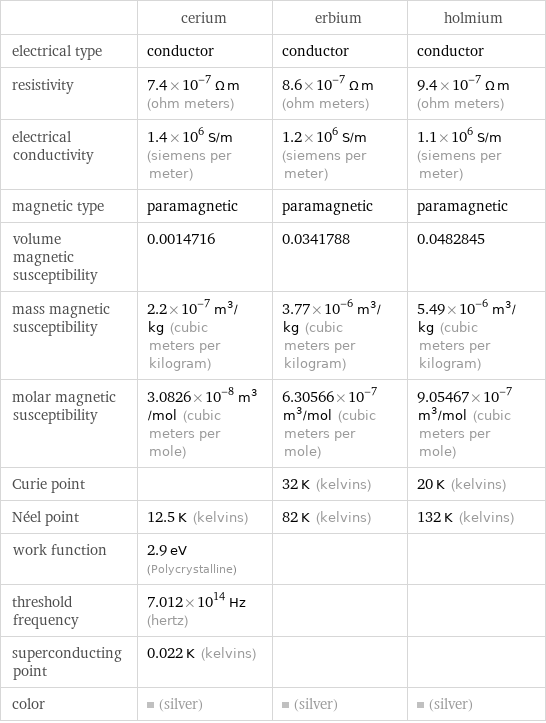
| cerium | erbium | holmium electrical type | conductor | conductor | conductor resistivity | 7.4×10^-7 Ω m (ohm meters) | 8.6×10^-7 Ω m (ohm meters) | 9.4×10^-7 Ω m (ohm meters) electrical conductivity | 1.4×10^6 S/m (siemens per meter) | 1.2×10^6 S/m (siemens per meter) | 1.1×10^6 S/m (siemens per meter) magnetic type | paramagnetic | paramagnetic | paramagnetic volume magnetic susceptibility | 0.0014716 | 0.0341788 | 0.0482845 mass magnetic susceptibility | 2.2×10^-7 m^3/kg (cubic meters per kilogram) | 3.77×10^-6 m^3/kg (cubic meters per kilogram) | 5.49×10^-6 m^3/kg (cubic meters per kilogram) molar magnetic susceptibility | 3.0826×10^-8 m^3/mol (cubic meters per mole) | 6.30566×10^-7 m^3/mol (cubic meters per mole) | 9.05467×10^-7 m^3/mol (cubic meters per mole) Curie point | | 32 K (kelvins) | 20 K (kelvins) Néel point | 12.5 K (kelvins) | 82 K (kelvins) | 132 K (kelvins) work function | 2.9 eV (Polycrystalline) | | threshold frequency | 7.012×10^14 Hz (hertz) | | superconducting point | 0.022 K (kelvins) | | color | (silver) | (silver) | (silver)
Reactivity
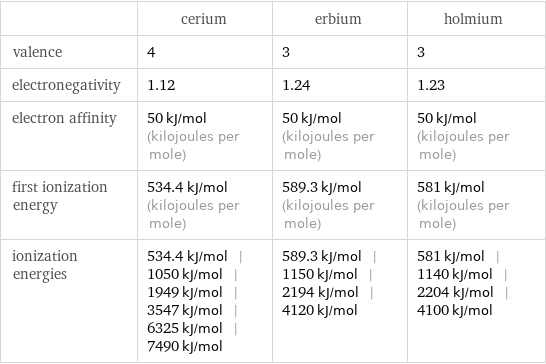
| cerium | erbium | holmium valence | 4 | 3 | 3 electronegativity | 1.12 | 1.24 | 1.23 electron affinity | 50 kJ/mol (kilojoules per mole) | 50 kJ/mol (kilojoules per mole) | 50 kJ/mol (kilojoules per mole) first ionization energy | 534.4 kJ/mol (kilojoules per mole) | 589.3 kJ/mol (kilojoules per mole) | 581 kJ/mol (kilojoules per mole) ionization energies | 534.4 kJ/mol | 1050 kJ/mol | 1949 kJ/mol | 3547 kJ/mol | 6325 kJ/mol | 7490 kJ/mol | 589.3 kJ/mol | 1150 kJ/mol | 2194 kJ/mol | 4120 kJ/mol | 581 kJ/mol | 1140 kJ/mol | 2204 kJ/mol | 4100 kJ/mol
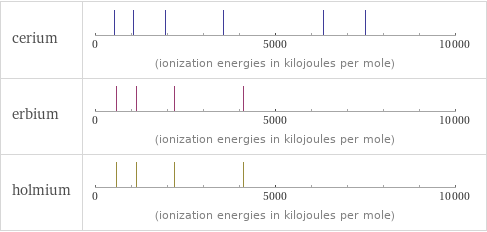
Reactivity
Atomic properties

| cerium | erbium | holmium term symbol | ^1G_4 | ^3H_6 | ^4I_(15/2) atomic radius | 185 pm | 175 pm | 175 pm covalent radius | 204 pm | 189 pm | 192 pm (electronic ground state properties)
Abundances
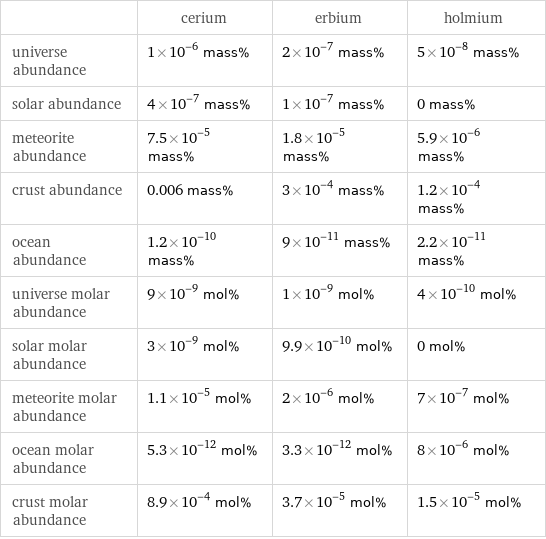
| cerium | erbium | holmium universe abundance | 1×10^-6 mass% | 2×10^-7 mass% | 5×10^-8 mass% solar abundance | 4×10^-7 mass% | 1×10^-7 mass% | 0 mass% meteorite abundance | 7.5×10^-5 mass% | 1.8×10^-5 mass% | 5.9×10^-6 mass% crust abundance | 0.006 mass% | 3×10^-4 mass% | 1.2×10^-4 mass% ocean abundance | 1.2×10^-10 mass% | 9×10^-11 mass% | 2.2×10^-11 mass% universe molar abundance | 9×10^-9 mol% | 1×10^-9 mol% | 4×10^-10 mol% solar molar abundance | 3×10^-9 mol% | 9.9×10^-10 mol% | 0 mol% meteorite molar abundance | 1.1×10^-5 mol% | 2×10^-6 mol% | 7×10^-7 mol% ocean molar abundance | 5.3×10^-12 mol% | 3.3×10^-12 mol% | 8×10^-6 mol% crust molar abundance | 8.9×10^-4 mol% | 3.7×10^-5 mol% | 1.5×10^-5 mol%