Input interpretation

inorganic salts | molar mass
Summary
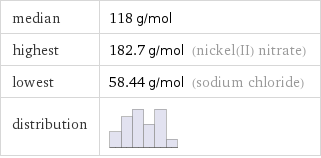
median | 118 g/mol highest | 182.7 g/mol (nickel(II) nitrate) lowest | 58.44 g/mol (sodium chloride) distribution |
Units

Distribution plots
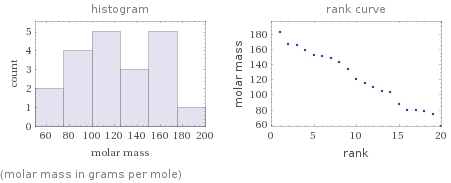
(molar mass in grams per mole)
Molar mass rankings
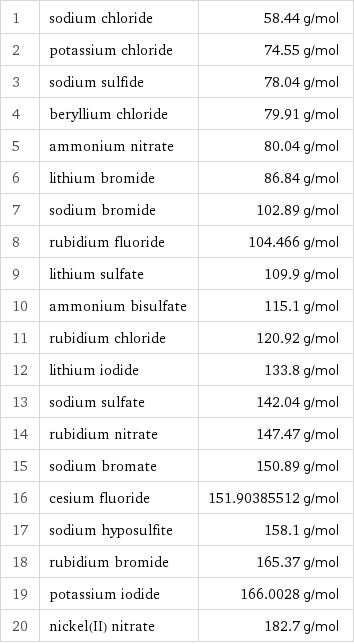
1 | sodium chloride | 58.44 g/mol 2 | potassium chloride | 74.55 g/mol 3 | sodium sulfide | 78.04 g/mol 4 | beryllium chloride | 79.91 g/mol 5 | ammonium nitrate | 80.04 g/mol 6 | lithium bromide | 86.84 g/mol 7 | sodium bromide | 102.89 g/mol 8 | rubidium fluoride | 104.466 g/mol 9 | lithium sulfate | 109.9 g/mol 10 | ammonium bisulfate | 115.1 g/mol 11 | rubidium chloride | 120.92 g/mol 12 | lithium iodide | 133.8 g/mol 13 | sodium sulfate | 142.04 g/mol 14 | rubidium nitrate | 147.47 g/mol 15 | sodium bromate | 150.89 g/mol 16 | cesium fluoride | 151.90385512 g/mol 17 | sodium hyposulfite | 158.1 g/mol 18 | rubidium bromide | 165.37 g/mol 19 | potassium iodide | 166.0028 g/mol 20 | nickel(II) nitrate | 182.7 g/mol
Unit conversion for median molar mass 118 g/mol
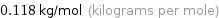
0.118 kg/mol (kilograms per mole)
Comparisons for median molar mass 118 g/mol
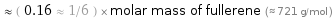
≈ ( 0.16 ≈ 1/6 ) × molar mass of fullerene (≈ 721 g/mol )
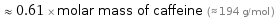
≈ 0.61 × molar mass of caffeine (≈ 194 g/mol )
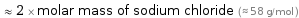
≈ 2 × molar mass of sodium chloride (≈ 58 g/mol )
Corresponding quantities

Mass of a molecule m from m = M/N_A: | 2×10^-22 grams | 2×10^-25 kg (kilograms) | 118 u (unified atomic mass units) | 118 Da (daltons)
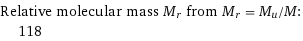
Relative molecular mass M_r from M_r = M_u/M: | 118