Input interpretation
polyatomic ions
Members

ammonium cation | hydronium cation | dimercury(I) cation | cyanide anion | hydrogen carbonate anion | hydrogen sulfate anion | hydroxide anion | nitrate anion | nitrite anion | perchlorate anion | permanganate anion | carbonate anion | chromate anion | dichromate anion | sulfate anion | sulfite anion | thiosulfate anion | hydrogen phosphate anion | phosphate anion | phosphite anion (total: 20)
Ionic radii
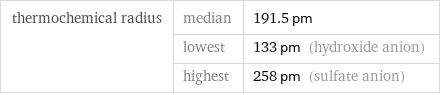
thermochemical radius | median | 191.5 pm | lowest | 133 pm (hydroxide anion) | highest | 258 pm (sulfate anion)
Units
