Input interpretation
silver(I) carbonate
Chemical names and formulas

formula | Ag_2CO_3 name | silver(I) carbonate IUPAC name | disilver; carbonate alternate names | carbonic acid, disilver(1+) salt | carbonic acid; silver | disilver carbonate | silver carbonate mass fractions | Ag (silver) 78.2% | C (carbon) 4.36% | O (oxygen) 17.4%
Structure diagram
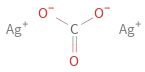
Structure diagram
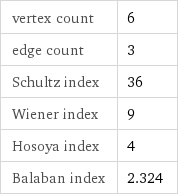
vertex count | 6 edge count | 3 Schultz index | 36 Wiener index | 9 Hosoya index | 4 Balaban index | 2.324
Basic properties
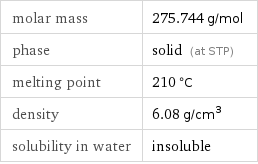
molar mass | 275.744 g/mol phase | solid (at STP) melting point | 210 °C density | 6.08 g/cm^3 solubility in water | insoluble
Units

Solid properties (at STP)

density | 6.08 g/cm^3
Units

Thermodynamic properties
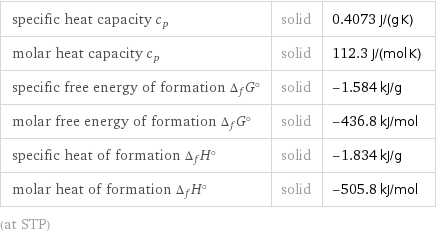
specific heat capacity c_p | solid | 0.4073 J/(g K) molar heat capacity c_p | solid | 112.3 J/(mol K) specific free energy of formation Δ_fG° | solid | -1.584 kJ/g molar free energy of formation Δ_fG° | solid | -436.8 kJ/mol specific heat of formation Δ_fH° | solid | -1.834 kJ/g molar heat of formation Δ_fH° | solid | -505.8 kJ/mol (at STP)
Chemical identifiers
![CAS number | 534-16-7 Beilstein number | 6936654 PubChem CID number | 92796 PubChem SID number | 24850822 SMILES identifier | C(=O)([O-])[O-].[Ag+].[Ag+] InChI identifier | InChI=1S/CH2O3.2Ag/c2-1(3)4;;/h(H2, 2, 3, 4);;/q;2*+1/p-2 RTECS number | FG0700000 MDL number | MFCD00003403](../image_source/805d24b49a627ec99bf8d2266d485475.png)
CAS number | 534-16-7 Beilstein number | 6936654 PubChem CID number | 92796 PubChem SID number | 24850822 SMILES identifier | C(=O)([O-])[O-].[Ag+].[Ag+] InChI identifier | InChI=1S/CH2O3.2Ag/c2-1(3)4;;/h(H2, 2, 3, 4);;/q;2*+1/p-2 RTECS number | FG0700000 MDL number | MFCD00003403
Toxicity properties

RTECS classes | other
Ion equivalents

(CO_3)^(2-) (carbonate anion) | 1 Ag^+ (silver(I) cation) | 2