Input interpretation
potassium dihydrogen phospho nate | hydrazine hydrate
Chemical names and formulas
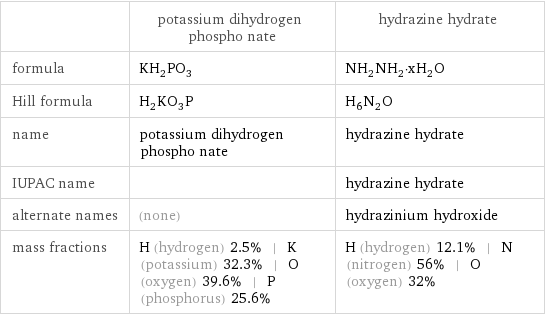
| potassium dihydrogen phospho nate | hydrazine hydrate formula | KH_2PO_3 | NH_2NH_2·xH_2O Hill formula | H_2KO_3P | H_6N_2O name | potassium dihydrogen phospho nate | hydrazine hydrate IUPAC name | | hydrazine hydrate alternate names | (none) | hydrazinium hydroxide mass fractions | H (hydrogen) 2.5% | K (potassium) 32.3% | O (oxygen) 39.6% | P (phosphorus) 25.6% | H (hydrogen) 12.1% | N (nitrogen) 56% | O (oxygen) 32%
Structure diagrams
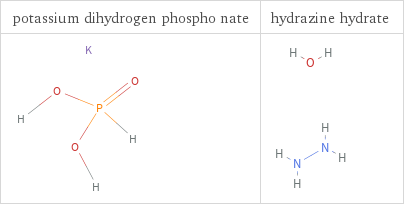
Structure diagrams
Basic properties

| potassium dihydrogen phospho nate | hydrazine hydrate molar mass | 121.09 g/mol | 50.06 g/mol density | | 1.029 g/cm^3
Units
