Input interpretation
methanol
Chemical names and formulas
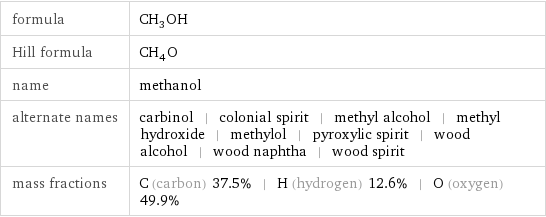
formula | CH_3OH Hill formula | CH_4O name | methanol alternate names | carbinol | colonial spirit | methyl alcohol | methyl hydroxide | methylol | pyroxylic spirit | wood alcohol | wood naphtha | wood spirit mass fractions | C (carbon) 37.5% | H (hydrogen) 12.6% | O (oxygen) 49.9%
Lewis structure

Draw the Lewis structure of methanol. Start by drawing the overall structure of the molecule: Count the total valence electrons of the carbon (n_C, val = 4), hydrogen (n_H, val = 1), and oxygen (n_O, val = 6) atoms: n_C, val + 4 n_H, val + n_O, val = 14 Calculate the number of electrons needed to completely fill the valence shells for carbon (n_C, full = 8), hydrogen (n_H, full = 2), and oxygen (n_O, full = 8): n_C, full + 4 n_H, full + n_O, full = 24 Subtracting these two numbers shows that 24 - 14 = 10 bonding electrons are needed. Each bond has two electrons, so the above diagram has all the necessary bonds. There are 5 bonds and hence 10 bonding electrons in the diagram. Lastly, fill in the remaining unbonded electrons on each atom. In total, there remain 14 - 10 = 4 electrons left to draw: Answer: | |
3D structure

3D structure
Basic properties
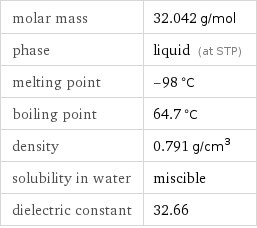
molar mass | 32.042 g/mol phase | liquid (at STP) melting point | -98 °C boiling point | 64.7 °C density | 0.791 g/cm^3 solubility in water | miscible dielectric constant | 32.66
Liquid properties (at STP)
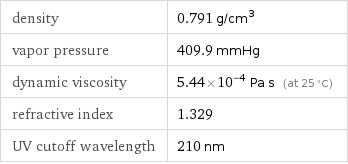
density | 0.791 g/cm^3 vapor pressure | 409.9 mmHg dynamic viscosity | 5.44×10^-4 Pa s (at 25 °C) refractive index | 1.329 UV cutoff wavelength | 210 nm
Units
Thermodynamic properties
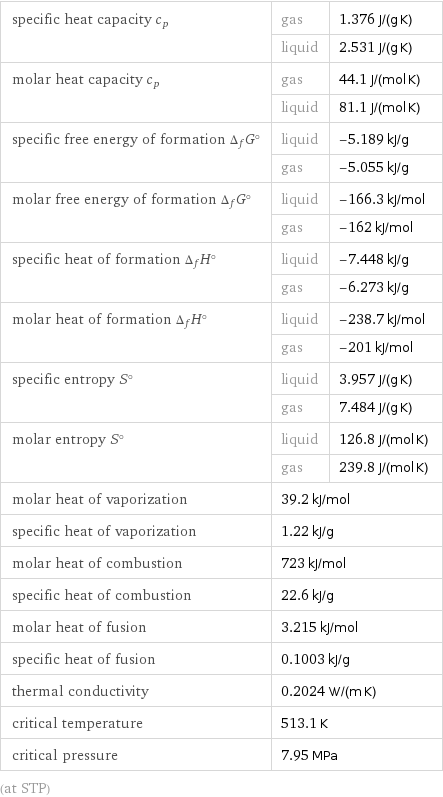
specific heat capacity c_p | gas | 1.376 J/(g K) | liquid | 2.531 J/(g K) molar heat capacity c_p | gas | 44.1 J/(mol K) | liquid | 81.1 J/(mol K) specific free energy of formation Δ_fG° | liquid | -5.189 kJ/g | gas | -5.055 kJ/g molar free energy of formation Δ_fG° | liquid | -166.3 kJ/mol | gas | -162 kJ/mol specific heat of formation Δ_fH° | liquid | -7.448 kJ/g | gas | -6.273 kJ/g molar heat of formation Δ_fH° | liquid | -238.7 kJ/mol | gas | -201 kJ/mol specific entropy S° | liquid | 3.957 J/(g K) | gas | 7.484 J/(g K) molar entropy S° | liquid | 126.8 J/(mol K) | gas | 239.8 J/(mol K) molar heat of vaporization | 39.2 kJ/mol | specific heat of vaporization | 1.22 kJ/g | molar heat of combustion | 723 kJ/mol | specific heat of combustion | 22.6 kJ/g | molar heat of fusion | 3.215 kJ/mol | specific heat of fusion | 0.1003 kJ/g | thermal conductivity | 0.2024 W/(m K) | critical temperature | 513.1 K | critical pressure | 7.95 MPa | (at STP)
Phase diagram
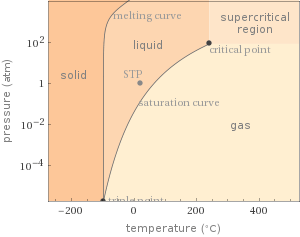
Phase diagram
Units

Chemical identifiers

CAS number | 67-56-1 Beilstein number | 1098229 PubChem CID number | 887 PubChem SID number | 24850836 SMILES identifier | CO InChI identifier | InChI=1/CH4O/c1-2/h2H, 1H3 RTECS number | PC1400000 MDL number | MFCD00004595
NFPA label

NFPA label

NFPA health rating | 1 NFPA fire rating | 3 NFPA reactivity rating | 0
Safety properties
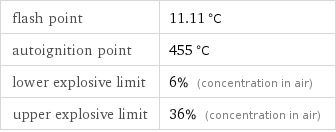
flash point | 11.11 °C autoignition point | 455 °C lower explosive limit | 6% (concentration in air) upper explosive limit | 36% (concentration in air)
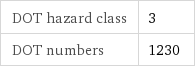
DOT hazard class | 3 DOT numbers | 1230
Toxicity properties
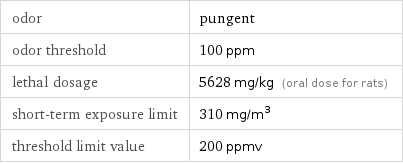
odor | pungent odor threshold | 100 ppm lethal dosage | 5628 mg/kg (oral dose for rats) short-term exposure limit | 310 mg/m^3 threshold limit value | 200 ppmv

probable lethal dose for man | 1 L (liter) long-term exposure limit | 250 mg/m^3 (over 8 hours) RTECS classes | drug | mutagen | reproductive effector | human data | primary irritant