Input interpretation

alkali metals | molar heat of fusion
Summary
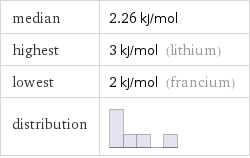
median | 2.26 kJ/mol highest | 3 kJ/mol (lithium) lowest | 2 kJ/mol (francium) distribution |
Units

Ranked values

| | visual | ratios | 6 | cesium | | 0.7 | 1 5 | rubidium | | 0.73 | 1.05 4 | francium | | 0.7 | 1 3 | potassium | | 0.78 | 1.11 2 | sodium | | 0.87 | 1.24 1 | lithium | | 1 | 1.4
Molar heat of fusion rankings

1 | francium | 2 kJ/mol 2 | cesium | 2.09 kJ/mol 3 | rubidium | 2.19 kJ/mol 4 | potassium | 2.33 kJ/mol 5 | sodium | 2.6 kJ/mol 6 | lithium | 3 kJ/mol
Unit conversions for median molar heat of fusion 2.26 kJ/mol
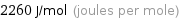
2260 J/mol (joules per mole)
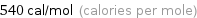
540 cal/mol (calories per mole)
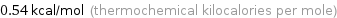
0.54 kcal/mol (thermochemical kilocalories per mole)

0.0234 eV (molar electronvolts)