Input interpretation

elements | Néel point
Summary

median | 100 K highest | 393 K (chromium) lowest | 12.5 K (cerium) distribution | | (based on 11 values; 107 unavailable)
Entities with missing values
hydrogen | helium | lithium | beryllium | boron | carbon | nitrogen | oxygen | fluorine | neon | ... (total: 107)
Distribution plots
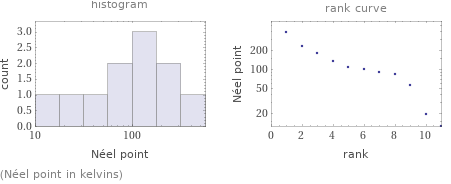
(Néel point in kelvins)
Néel point rankings
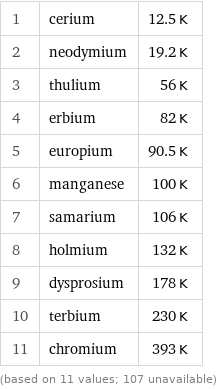
1 | cerium | 12.5 K 2 | neodymium | 19.2 K 3 | thulium | 56 K 4 | erbium | 82 K 5 | europium | 90.5 K 6 | manganese | 100 K 7 | samarium | 106 K 8 | holmium | 132 K 9 | dysprosium | 178 K 10 | terbium | 230 K 11 | chromium | 393 K (based on 11 values; 107 unavailable)
Unit conversions for median Néel point 100 K

-173.15 °C (degrees Celsius)
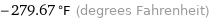
-279.67 °F (degrees Fahrenheit)

180 °R (degrees Rankine)

-138.52 °Ré (degrees Réaumur)
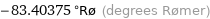
-83.40375 °Rø (degrees Rømer)
Comparison for median Néel point 100 K
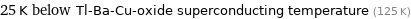
25 K below Tl-Ba-Cu-oxide superconducting temperature (125 K)
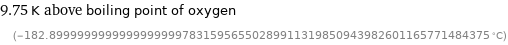
9.75 K above boiling point of oxygen (-182.899999999999999999978315956550289911319850943982601165771484375 °C)
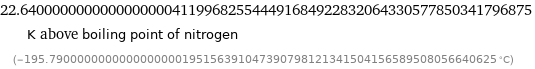
22.6400000000000000000411996825544491684922832064330577850341796875 K above boiling point of nitrogen (-195.7900000000000000000195156391047390798121341504156589508056640625 °C)
Corresponding quantities
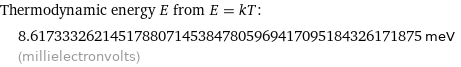
Thermodynamic energy E from E = kT: | 8.6173332621451788071453847805969417095184326171875 meV (millielectronvolts)
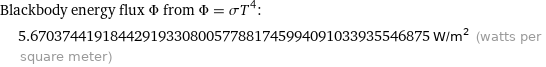
Blackbody energy flux Φ from Φ = σT^4: | 5.67037441918442919330800577881745994091033935546875 W/m^2 (watts per square meter)
Thermodynamic properties
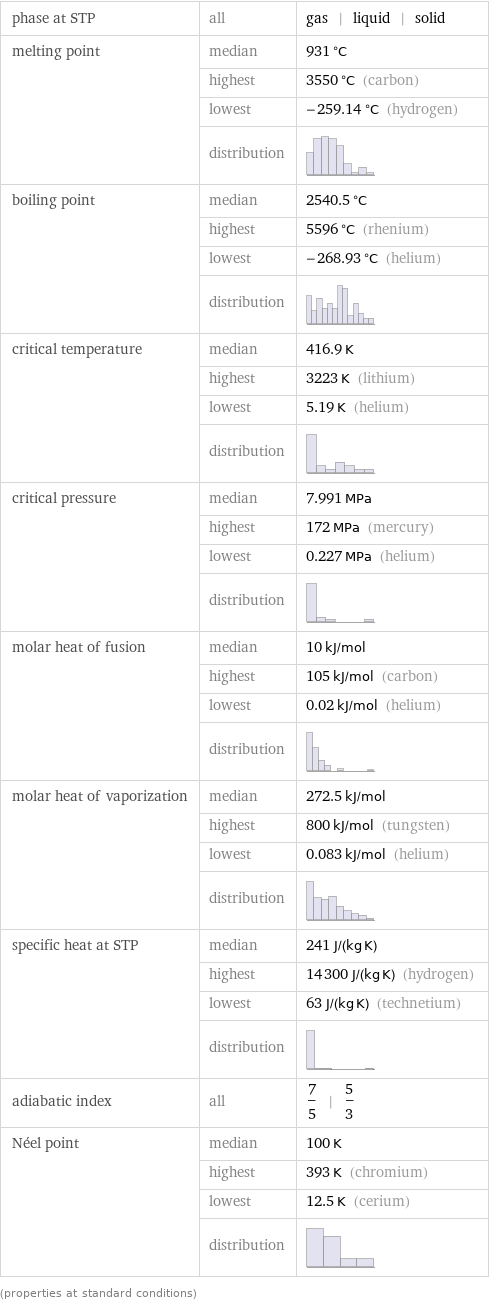
phase at STP | all | gas | liquid | solid melting point | median | 931 °C | highest | 3550 °C (carbon) | lowest | -259.14 °C (hydrogen) | distribution | boiling point | median | 2540.5 °C | highest | 5596 °C (rhenium) | lowest | -268.93 °C (helium) | distribution | critical temperature | median | 416.9 K | highest | 3223 K (lithium) | lowest | 5.19 K (helium) | distribution | critical pressure | median | 7.991 MPa | highest | 172 MPa (mercury) | lowest | 0.227 MPa (helium) | distribution | molar heat of fusion | median | 10 kJ/mol | highest | 105 kJ/mol (carbon) | lowest | 0.02 kJ/mol (helium) | distribution | molar heat of vaporization | median | 272.5 kJ/mol | highest | 800 kJ/mol (tungsten) | lowest | 0.083 kJ/mol (helium) | distribution | specific heat at STP | median | 241 J/(kg K) | highest | 14300 J/(kg K) (hydrogen) | lowest | 63 J/(kg K) (technetium) | distribution | adiabatic index | all | 7/5 | 5/3 Néel point | median | 100 K | highest | 393 K (chromium) | lowest | 12.5 K (cerium) | distribution | (properties at standard conditions)