Input interpretation

alkaline earth metals | first molar ionization energy
Summary

median | 569.6 kJ/mol highest | 899.5 kJ/mol (beryllium) lowest | 502.9 kJ/mol (barium) distribution |
Units

Ranked values
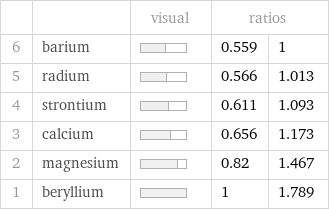
| | visual | ratios | 6 | barium | | 0.559 | 1 5 | radium | | 0.566 | 1.013 4 | strontium | | 0.611 | 1.093 3 | calcium | | 0.656 | 1.173 2 | magnesium | | 0.82 | 1.467 1 | beryllium | | 1 | 1.789
First molar ionization energy rankings
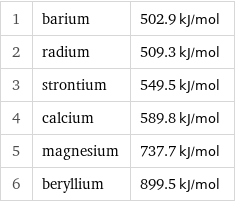
1 | barium | 502.9 kJ/mol 2 | radium | 509.3 kJ/mol 3 | strontium | 549.5 kJ/mol 4 | calcium | 589.8 kJ/mol 5 | magnesium | 737.7 kJ/mol 6 | beryllium | 899.5 kJ/mol
Unit conversions for median first molar ionization energy 569.6 kJ/mol
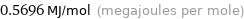
0.5696 MJ/mol (megajoules per mole)
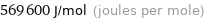
569600 J/mol (joules per mole)
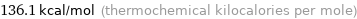
136.1 kcal/mol (thermochemical kilocalories per mole)

5.904 eV (molar electronvolts)
Comparisons for median first molar ionization energy 569.6 kJ/mol

≈ ( 0.24 ≈ 1/4 ) × molar energy required to remove the first electron from helium ( 2372 kJ/mol )
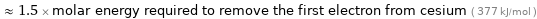
≈ 1.5 × molar energy required to remove the first electron from cesium ( 377 kJ/mol )