Input interpretation

s-block elements | atomic radius
Summary
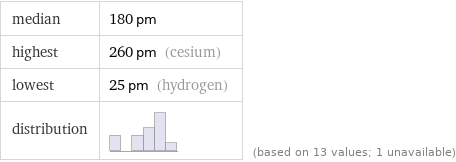
median | 180 pm highest | 260 pm (cesium) lowest | 25 pm (hydrogen) distribution | | (based on 13 values; 1 unavailable)
Entity with missing value
francium
Plot
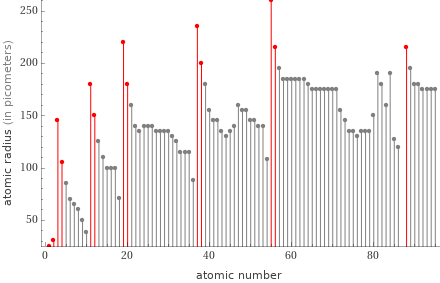
Plot
Periodic table location
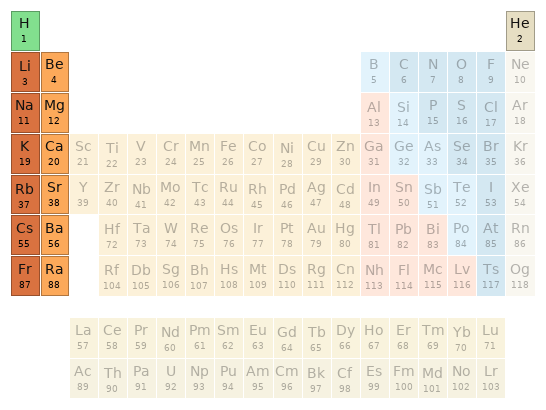
Periodic table location
Atomic radii properties

atomic radius (empirical) | median | 190 pm | highest | 260 pm (cesium) | lowest | 25 pm (hydrogen) | distribution | atomic radius (calculated) | median | 192 pm | highest | 298 pm (cesium) | lowest | 31 pm (helium) | distribution | covalent radius | median | 186 pm | highest | 260 pm (francium) | lowest | 28 pm (helium) | distribution | van der Waals radius | median | 178 pm | highest | 275 pm (potassium) | lowest | 120 pm (hydrogen) | distribution |
Units

Distribution plots
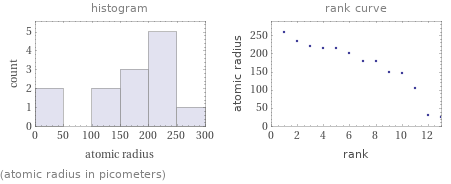
(atomic radius in picometers)
Atomic radius rankings

1 | hydrogen | 25 pm 2 | helium | 31 pm 3 | beryllium | 105 pm 4 | lithium | 145 pm 5 | magnesium | 150 pm 6 | calcium | 180 pm 7 | sodium | 180 pm 8 | strontium | 200 pm 9 | radium | 215 pm 10 | barium | 215 pm 11 | potassium | 220 pm 12 | rubidium | 235 pm 13 | cesium | 260 pm (based on 13 values; 1 unavailable)
Unit conversions for median atomic radius 180 pm
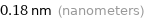
0.18 nm (nanometers)
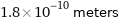
1.8×10^-10 meters
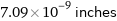
7.09×10^-9 inches
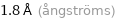
1.8 Å (ångströms)
Comparisons for median atomic radius 180 pm
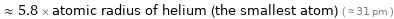
≈ 5.8 × atomic radius of helium (the smallest atom) ( ≈ 31 pm )
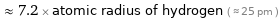
≈ 7.2 × atomic radius of hydrogen ( ≈ 25 pm )