Input interpretation
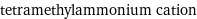
tetramethylammonium cation
Lewis structure

Draw the Lewis structure of tetramethylammonium cation. Start by drawing the overall structure of the molecule: Count the total valence electrons of the carbon (n_C, val = 4), hydrogen (n_H, val = 1), and nitrogen (n_N, val = 5) atoms, including the net charge: 4 n_C, val + 12 n_H, val + n_N, val - n_charge = 32 Calculate the number of electrons needed to completely fill the valence shells for carbon (n_C, full = 8), hydrogen (n_H, full = 2), and nitrogen (n_N, full = 8): 4 n_C, full + 12 n_H, full + n_N, full = 64 Subtracting these two numbers shows that 64 - 32 = 32 bonding electrons are needed. Each bond has two electrons, so the above diagram has all the necessary bonds. There are 16 bonds and hence 32 bonding electrons in the diagram. Fill in the remaining unbonded electrons on each atom. In total, there remain 32 - 32 = 0 electrons left to draw. Lastly, fill in the formal charges: Answer: | |
General properties
![formula | ([N(CH_3)_4])^+ net ionic charge | +1 alternate names | tetramethylazanium | tetramethylammonium(1+)](../image_source/07f49ab6d919225b7e031ee198fa926f.png)
formula | ([N(CH_3)_4])^+ net ionic charge | +1 alternate names | tetramethylazanium | tetramethylammonium(1+)
Ionic radius

thermochemical radius | 201 pm
Units

Other properties

ion class | cations | onium ions | polyatomic ions