Input interpretation

Arrhenius bases | melting point
Summary

median | 372 °C highest | 2580 °C (lime) lowest | -57.5 °C (ammonium hydroxide) distribution |
Distribution plots
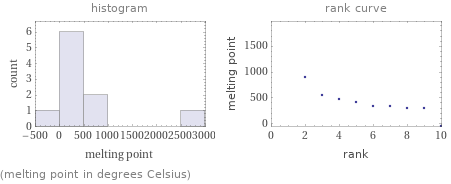
(melting point in degrees Celsius)
Melting point rankings
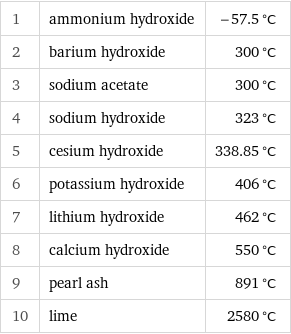
1 | ammonium hydroxide | -57.5 °C 2 | barium hydroxide | 300 °C 3 | sodium acetate | 300 °C 4 | sodium hydroxide | 323 °C 5 | cesium hydroxide | 338.85 °C 6 | potassium hydroxide | 406 °C 7 | lithium hydroxide | 462 °C 8 | calcium hydroxide | 550 °C 9 | pearl ash | 891 °C 10 | lime | 2580 °C
Unit conversions for median melting point 372 °C
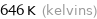
646 K (kelvins)
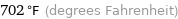
702 °F (degrees Fahrenheit)
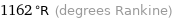
1162 °R (degrees Rankine)

298 °Ré (degrees Réaumur)
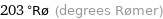
203 °Rø (degrees Rømer)
Comparison for median melting point 372 °C
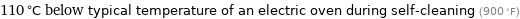
110 °C below typical temperature of an electric oven during self-cleaning (900 °F)
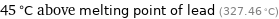
45 °C above melting point of lead (327.46 °C)
Corresponding quantities

Thermodynamic energy E from E = kT: | 56 meV (millielectronvolts)
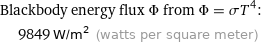
Blackbody energy flux Φ from Φ = σT^4: | 9849 W/m^2 (watts per square meter)

Approximate luminous exitance from a planar blackbody radiator perpendicular to its surface: | 2.9×10^-5 lx (lux)