Input interpretation
cerussete
Chemical names and formulas

formula | PbCO_3 Hill formula | CO_3Pb name | cerussete IUPAC name | lead(+2) cation carbonate alternate names | carbonic acid, lead(2+) salt | cerussite | lead carbonate mass fractions | C (carbon) 4.5% | O (oxygen) 18% | Pb (lead) 77.5%
Structure diagram
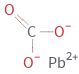
Structure diagram
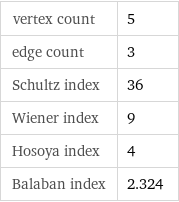
vertex count | 5 edge count | 3 Schultz index | 36 Wiener index | 9 Hosoya index | 4 Balaban index | 2.324
Basic properties
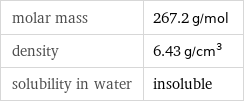
molar mass | 267.2 g/mol density | 6.43 g/cm^3 solubility in water | insoluble
Units

Thermodynamic properties
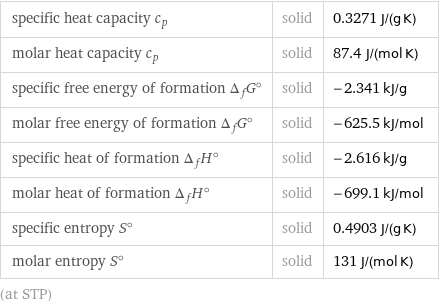
specific heat capacity c_p | solid | 0.3271 J/(g K) molar heat capacity c_p | solid | 87.4 J/(mol K) specific free energy of formation Δ_fG° | solid | -2.341 kJ/g molar free energy of formation Δ_fG° | solid | -625.5 kJ/mol specific heat of formation Δ_fH° | solid | -2.616 kJ/g molar heat of formation Δ_fH° | solid | -699.1 kJ/mol specific entropy S° | solid | 0.4903 J/(g K) molar entropy S° | solid | 131 J/(mol K) (at STP)
Chemical identifiers
![CAS number | 25510-11-6 Beilstein number | 8591 PubChem CID number | 11727 SMILES identifier | C(=O)([O-])[O-].[Pb+2] InChI identifier | InChI=1/CH2O3.Pb/c2-1(3)4;/h(H2, 2, 3, 4);/q;+2/p-2/fCO3.Pb/q-2;m EU number | 247-054-3](../image_source/66ea77fdd18175a06c5adacb089ea3df.png)
CAS number | 25510-11-6 Beilstein number | 8591 PubChem CID number | 11727 SMILES identifier | C(=O)([O-])[O-].[Pb+2] InChI identifier | InChI=1/CH2O3.Pb/c2-1(3)4;/h(H2, 2, 3, 4);/q;+2/p-2/fCO3.Pb/q-2;m EU number | 247-054-3
Ion equivalents

Pb^(2+) (lead(II) cation) | 1 (CO_3)^(2-) (carbonate anion) | 1