Input interpretation

p block ions | ionic radius
Summary

median | 99 pm highest | 220 pm (iodide anion) lowest | 46 pm (arsenic(V) cation) distribution | | (based on 16 values; 4 unavailable)
Entities with missing values
helium cation | nitride anion | phosphide anion | arsenide anion (total: 4)
Distribution plots
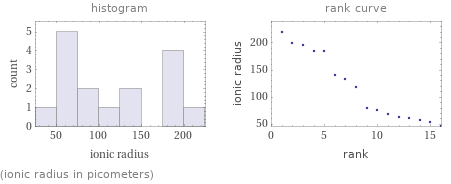
(ionic radius in picometers)
Ionic radius rankings
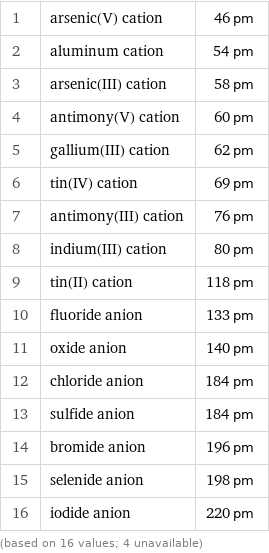
1 | arsenic(V) cation | 46 pm 2 | aluminum cation | 54 pm 3 | arsenic(III) cation | 58 pm 4 | antimony(V) cation | 60 pm 5 | gallium(III) cation | 62 pm 6 | tin(IV) cation | 69 pm 7 | antimony(III) cation | 76 pm 8 | indium(III) cation | 80 pm 9 | tin(II) cation | 118 pm 10 | fluoride anion | 133 pm 11 | oxide anion | 140 pm 12 | chloride anion | 184 pm 13 | sulfide anion | 184 pm 14 | bromide anion | 196 pm 15 | selenide anion | 198 pm 16 | iodide anion | 220 pm (based on 16 values; 4 unavailable)
Ionic radii
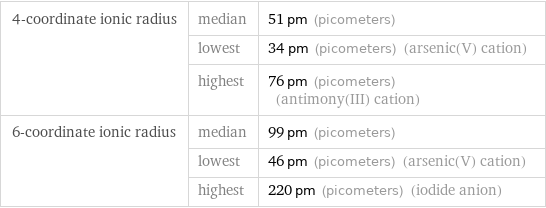
4-coordinate ionic radius | median | 51 pm (picometers) | lowest | 34 pm (picometers) (arsenic(V) cation) | highest | 76 pm (picometers) (antimony(III) cation) 6-coordinate ionic radius | median | 99 pm (picometers) | lowest | 46 pm (picometers) (arsenic(V) cation) | highest | 220 pm (picometers) (iodide anion)
Unit conversions for median ionic radius 99 pm

99/1000 nm (nanometers)
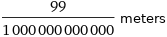
99/1000000000000 meters
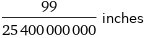
99/25400000000 inches
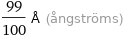
99/100 Å (ångströms)