Input interpretation
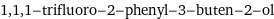
1, 1, 1-trifluoro-2-phenyl-3-buten-2-ol
Chemical names and formulas

formula | CH_2=CHC(CF_3)(OH)C_6H_5 Hill formula | C_10H_9F_3O name | 1, 1, 1-trifluoro-2-phenyl-3-buten-2-ol IUPAC name | 1, 1, 1-trifluoro-2-phenylbut-3-en-2-ol alternate names | 1, 1, 1-trifluoro-2-phenyl-but-3-en-2-ol | 1, 1, 1-trifluoro-2-phenylbut-3-en-2-ol | 2-phenyl-1, 1, 1-trifluoro-3-buten-2-ol mass fractions | C (carbon) 59.4% | F (fluorine) 28.2% | H (hydrogen) 4.49% | O (oxygen) 7.91%
Lewis structure
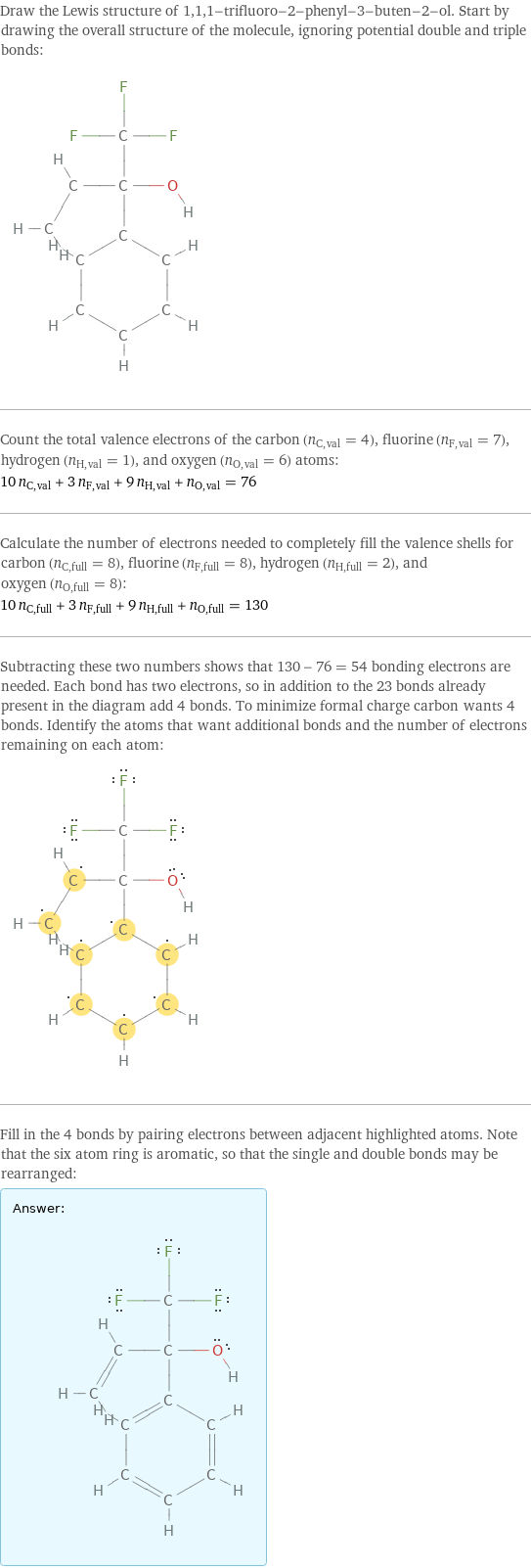
Draw the Lewis structure of 1, 1, 1-trifluoro-2-phenyl-3-buten-2-ol. Start by drawing the overall structure of the molecule, ignoring potential double and triple bonds: Count the total valence electrons of the carbon (n_C, val = 4), fluorine (n_F, val = 7), hydrogen (n_H, val = 1), and oxygen (n_O, val = 6) atoms: 10 n_C, val + 3 n_F, val + 9 n_H, val + n_O, val = 76 Calculate the number of electrons needed to completely fill the valence shells for carbon (n_C, full = 8), fluorine (n_F, full = 8), hydrogen (n_H, full = 2), and oxygen (n_O, full = 8): 10 n_C, full + 3 n_F, full + 9 n_H, full + n_O, full = 130 Subtracting these two numbers shows that 130 - 76 = 54 bonding electrons are needed. Each bond has two electrons, so in addition to the 23 bonds already present in the diagram add 4 bonds. To minimize formal charge carbon wants 4 bonds. Identify the atoms that want additional bonds and the number of electrons remaining on each atom: Fill in the 4 bonds by pairing electrons between adjacent highlighted atoms. Note that the six atom ring is aromatic, so that the single and double bonds may be rearranged: Answer: | |
3D structure
3D structure
Basic properties

molar mass | 202.18 g/mol phase | liquid (at STP) boiling point | 89.5 °C (measured at 1600 Pa) density | 1.22 g/cm^3
Units

Liquid properties (at STP)

density | 1.22 g/cm^3 refractive index | 1.474
Units

Chemical identifiers

CAS number | 134418-70-5 PubChem CID number | 2734576 PubChem SID number | 24879560 SMILES identifier | C=CC(C1=CC=CC=C1)(C(F)(F)F)O InChI identifier | InChI=1/C10H9F3O/c1-2-9(14, 10(11, 12)13)8-6-4-3-5-7-8/h2-7, 14H, 1H2 MDL number | MFCD03427175
Safety properties

flash point | 85 °C