Input interpretation

p block ions | net ionic charge
Table
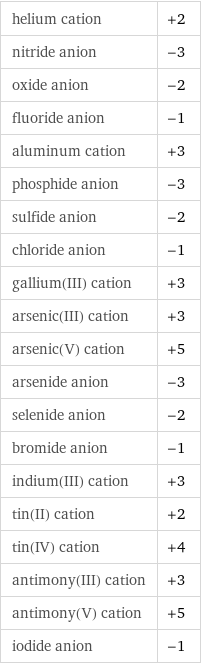
helium cation | +2 nitride anion | -3 oxide anion | -2 fluoride anion | -1 aluminum cation | +3 phosphide anion | -3 sulfide anion | -2 chloride anion | -1 gallium(III) cation | +3 arsenic(III) cation | +3 arsenic(V) cation | +5 arsenide anion | -3 selenide anion | -2 bromide anion | -1 indium(III) cation | +3 tin(II) cation | +2 tin(IV) cation | +4 antimony(III) cation | +3 antimony(V) cation | +5 iodide anion | -1