Input interpretation
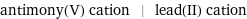
antimony(V) cation | lead(II) cation
Structure diagrams

Structure diagrams
General properties

| antimony(V) cation | lead(II) cation formula | Sb^(5+) | Pb^(2+) net ionic charge | +5 | +2 alternate names | antimony(V) | antimony(5+) | plumbous | lead(II) | lead(2+)
Ionic radii

| antimony(V) cation | lead(II) cation 6-coordinate ionic radius | 60 pm | 119 pm 8-coordinate ionic radius | | 129 pm 10-coordinate ionic radius | | 140 pm 12-coordinate ionic radius | | 149 pm
Units

Other properties

| antimony(V) cation | lead(II) cation ion class | antimony(V) ions | cations | group 15 ions | p block ions | biomolecule ions | cations | group 14 ions | lead(II) ions | p block ions common sources of ion | | lead(II) zirconate (1 eq) | lead(II) titanate (1 eq) | lead(II) salicylate (1 eq) | lead(II) perchlorate hydrate (1 eq) | lead(II) nitrate (1 eq) | lead(II) molybdate (1 eq) | lead(II) chromate (1 eq)