Input interpretation

chromium | work function
Results
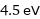
4.5 eV
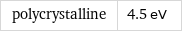
polycrystalline | 4.5 eV
Threshold frequency
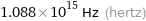
1.088×10^15 Hz (hertz)
Electromagnetic frequency range

NUV (near ultraviolet) | UVC
Unit conversion
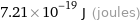
7.21×10^-19 J (joules)
Comparisons as energy
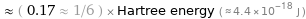
≈ ( 0.17 ≈ 1/6 ) × Hartree energy ( ≈ 4.4×10^-18 J )
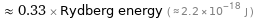
≈ 0.33 × Rydberg energy ( ≈ 2.2×10^-18 J )
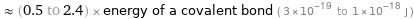
≈ (0.5 to 2.4) × energy of a covalent bond ( 3×10^-19 to 1×10^-18 J )
Corresponding quantities

Thermodynamic temperature T from E = kT: | 52220 K (kelvins)
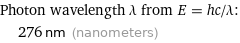
Photon wavelength λ from E = hc/λ: | 276 nm (nanometers)
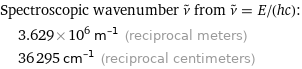
Spectroscopic wavenumber ν^~ from ν^~ = E/(hc): | 3.629×10^6 m^(-1) (reciprocal meters) | 36295 cm^(-1) (reciprocal centimeters)