Input interpretation

polyprotic acids | boiling point
Summary

median | 310 °C highest | 614 °C (edetic acid) lowest | -60 °C (hydrogen sulfide) distribution | | (based on 11 values; 9 unavailable)
Entities with missing values

chromic acid | pyrophosphoric acid | selenic acid | sulfurous acid | phosphorous acid | hydrogen selenide | tellurous acid | carbonic acid | boric acid (total: 9)
Distribution plots
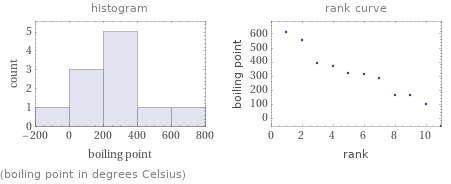
(boiling point in degrees Celsius)
Boiling point rankings
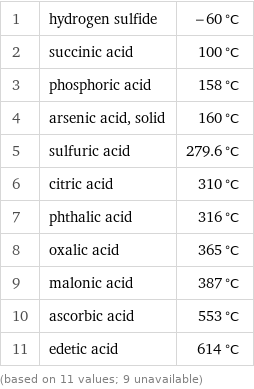
1 | hydrogen sulfide | -60 °C 2 | succinic acid | 100 °C 3 | phosphoric acid | 158 °C 4 | arsenic acid, solid | 160 °C 5 | sulfuric acid | 279.6 °C 6 | citric acid | 310 °C 7 | phthalic acid | 316 °C 8 | oxalic acid | 365 °C 9 | malonic acid | 387 °C 10 | ascorbic acid | 553 °C 11 | edetic acid | 614 °C (based on 11 values; 9 unavailable)
Unit conversions for median boiling point 310 °C
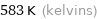
583 K (kelvins)
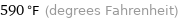
590 °F (degrees Fahrenheit)
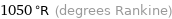
1050 °R (degrees Rankine)

248 °Ré (degrees Réaumur)
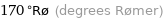
170 °Rø (degrees Rømer)
Comparison for median boiling point 310 °C
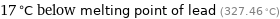
17 °C below melting point of lead (327.46 °C)

77 °C above autoignition temperature of book paper in Ray Bradbury's famous novel (451 °F)

(60 to 90) °C above autoignition temperature of paper (218 to 246 °C)
Corresponding quantities

Thermodynamic energy E from E = kT: | 50 meV (millielectronvolts)
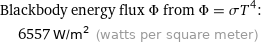
Blackbody energy flux Φ from Φ = σT^4: | 6557 W/m^2 (watts per square meter)

Approximate luminous exitance from a planar blackbody radiator perpendicular to its surface: | 7.2×10^-7 lx (lux)