Input interpretation
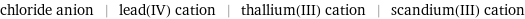
chloride anion | lead(IV) cation | thallium(III) cation | scandium(III) cation
Structure diagrams
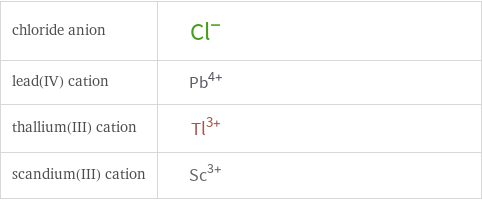
Structure diagrams
Ionic radii
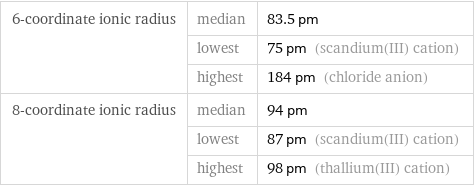
6-coordinate ionic radius | median | 83.5 pm | lowest | 75 pm (scandium(III) cation) | highest | 184 pm (chloride anion) 8-coordinate ionic radius | median | 94 pm | lowest | 87 pm (scandium(III) cation) | highest | 98 pm (thallium(III) cation)
Units
