Input interpretation
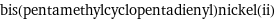
bis(pentamethylcyclopentadienyl)nickel(ii)
Basic properties
![molar mass | 329.1 g/mol formula | C_20H_30Ni empirical formula | C_20Ni_H_30 SMILES identifier | CC1=C(C)C(C)(C(=C1C)C)[Ni]C2(C)C(=C(C)C(=C2C)C)C InChI identifier | InChI=1/2C10H15.Ni/c2*1-6-7(2)9(4)10(5)8(6)3;/h2*1-5H3; InChI key | IBWGRWRPKIAFEV-UHFFFAOYSA-N](../image_source/4508833300525f4bb6ffe54aebb66e7b.png)
molar mass | 329.1 g/mol formula | C_20H_30Ni empirical formula | C_20Ni_H_30 SMILES identifier | CC1=C(C)C(C)(C(=C1C)C)[Ni]C2(C)C(=C(C)C(=C2C)C)C InChI identifier | InChI=1/2C10H15.Ni/c2*1-6-7(2)9(4)10(5)8(6)3;/h2*1-5H3; InChI key | IBWGRWRPKIAFEV-UHFFFAOYSA-N
Structure diagram
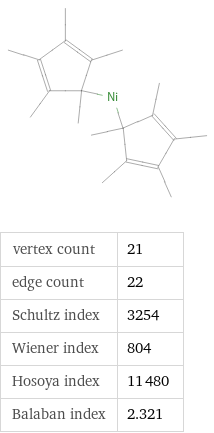
vertex count | 21 edge count | 22 Schultz index | 3254 Wiener index | 804 Hosoya index | 11480 Balaban index | 2.321
Quantitative molecular descriptors
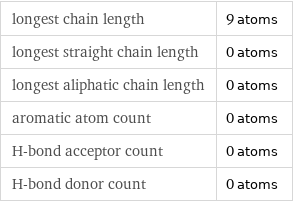
longest chain length | 9 atoms longest straight chain length | 0 atoms longest aliphatic chain length | 0 atoms aromatic atom count | 0 atoms H-bond acceptor count | 0 atoms H-bond donor count | 0 atoms
Elemental composition

Find the elemental composition for bis(pentamethylcyclopentadienyl)nickel(ii) in terms of the atom and mass percents: atom percent = N_i/N_atoms × 100% mass percent = (N_im_i)/m × 100% Plan: • Write the chemical formula and gather atomic masses from the periodic table. • Determine values for N_i, m_i, N_atoms and m using these items. • Finally, compute the percents and check the results. Write the chemical formula: C_20H_30Ni Use the chemical formula to count the number of atoms, N_i, for each element and find the total number of atoms, N_atoms, per molecule: | number of atoms C (carbon) | 20 Ni (nickel) | 1 H (hydrogen) | 30 N_atoms = 20 + 1 + 30 = 51 Divide each N_i by N_atoms to calculate atom fractions. Then use the property that atom fractions must sum to one to check the work: | number of atoms | atom fraction C (carbon) | 20 | 20/51 Ni (nickel) | 1 | 1/51 H (hydrogen) | 30 | 30/51 Check: 20/51 + 1/51 + 30/51 = 1 Compute atom percents using the atom fractions: | number of atoms | atom percent C (carbon) | 20 | 20/51 × 100% = 39.2% Ni (nickel) | 1 | 1/51 × 100% = 1.96% H (hydrogen) | 30 | 30/51 × 100% = 58.8% Look up the atomic mass, m_i, in unified atomic mass units, u, for each element in the periodic table: | number of atoms | atom percent | atomic mass/u C (carbon) | 20 | 39.2% | 12.011 Ni (nickel) | 1 | 1.96% | 58.6934 H (hydrogen) | 30 | 58.8% | 1.008 Multiply N_i by m_i to compute the mass for each element. Then sum those values to compute the molecular mass, m: | number of atoms | atom percent | atomic mass/u | mass/u C (carbon) | 20 | 39.2% | 12.011 | 20 × 12.011 = 240.220 Ni (nickel) | 1 | 1.96% | 58.6934 | 1 × 58.6934 = 58.6934 H (hydrogen) | 30 | 58.8% | 1.008 | 30 × 1.008 = 30.240 m = 240.220 u + 58.6934 u + 30.240 u = 329.1534 u Divide the mass for each element by m to calculate mass fractions. Then use the property that mass fractions must sum to one to check the work: | number of atoms | atom percent | mass fraction C (carbon) | 20 | 39.2% | 240.220/329.1534 Ni (nickel) | 1 | 1.96% | 58.6934/329.1534 H (hydrogen) | 30 | 58.8% | 30.240/329.1534 Check: 240.220/329.1534 + 58.6934/329.1534 + 30.240/329.1534 = 1 Compute mass percents using the mass fractions: Answer: | | | number of atoms | atom percent | mass percent C (carbon) | 20 | 39.2% | 240.220/329.1534 × 100% = 72.98% Ni (nickel) | 1 | 1.96% | 58.6934/329.1534 × 100% = 17.83% H (hydrogen) | 30 | 58.8% | 30.240/329.1534 × 100% = 9.187%
Elemental oxidation states
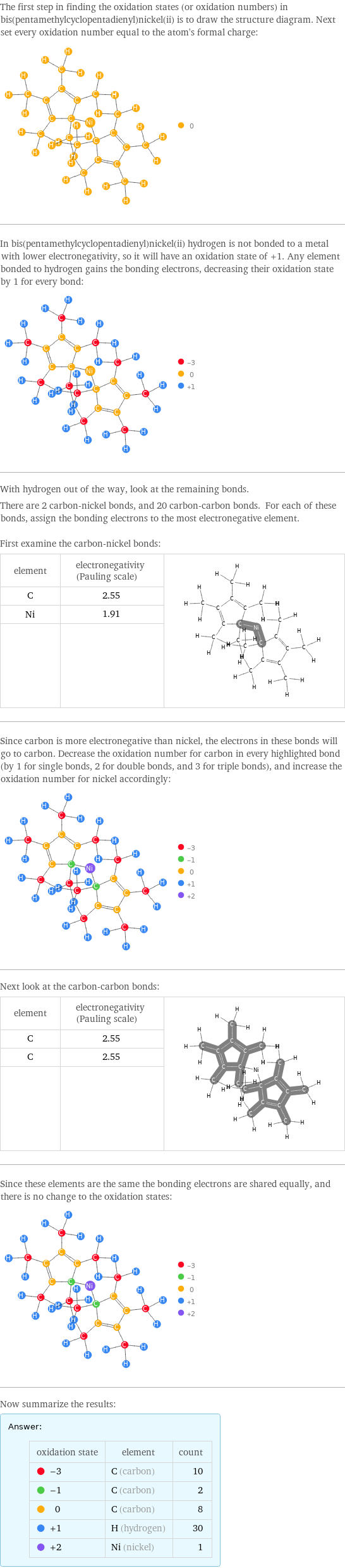
The first step in finding the oxidation states (or oxidation numbers) in bis(pentamethylcyclopentadienyl)nickel(ii) is to draw the structure diagram. Next set every oxidation number equal to the atom's formal charge: In bis(pentamethylcyclopentadienyl)nickel(ii) hydrogen is not bonded to a metal with lower electronegativity, so it will have an oxidation state of +1. Any element bonded to hydrogen gains the bonding electrons, decreasing their oxidation state by 1 for every bond: With hydrogen out of the way, look at the remaining bonds. There are 2 carbon-nickel bonds, and 20 carbon-carbon bonds. For each of these bonds, assign the bonding electrons to the most electronegative element. First examine the carbon-nickel bonds: element | electronegativity (Pauling scale) | C | 2.55 | Ni | 1.91 | | | Since carbon is more electronegative than nickel, the electrons in these bonds will go to carbon. Decrease the oxidation number for carbon in every highlighted bond (by 1 for single bonds, 2 for double bonds, and 3 for triple bonds), and increase the oxidation number for nickel accordingly: Next look at the carbon-carbon bonds: element | electronegativity (Pauling scale) | C | 2.55 | C | 2.55 | | | Since these elements are the same the bonding electrons are shared equally, and there is no change to the oxidation states: Now summarize the results: Answer: | | oxidation state | element | count -3 | C (carbon) | 10 -1 | C (carbon) | 2 0 | C (carbon) | 8 +1 | H (hydrogen) | 30 +2 | Ni (nickel) | 1
Topological indices
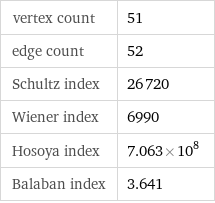
vertex count | 51 edge count | 52 Schultz index | 26720 Wiener index | 6990 Hosoya index | 7.063×10^8 Balaban index | 3.641