Input interpretation
gallium fluoride
Chemical names and formulas

formula | GaF_3 Hill formula | F_3Ga name | gallium fluoride IUPAC name | trifluorogallane alternate names | gallium fluoride trihydrate | gallium(III) fluoride | gallium trifluoride | trifluorogallane mass fractions | F (fluorine) 45% | Ga (gallium) 55%
Structure diagram
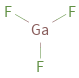
Structure diagram
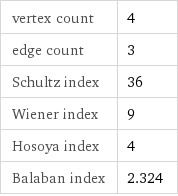
vertex count | 4 edge count | 3 Schultz index | 36 Wiener index | 9 Hosoya index | 4 Balaban index | 2.324
Basic properties
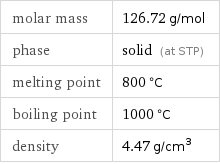
molar mass | 126.72 g/mol phase | solid (at STP) melting point | 800 °C boiling point | 1000 °C density | 4.47 g/cm^3
Units

Solid properties (at STP)

density | 4.47 g/cm^3
Units

Thermodynamic properties

specific free energy of formation Δ_fG° | solid | -8.565 kJ/g molar free energy of formation Δ_fG° | solid | -1085 kJ/mol specific heat of formation Δ_fH° | gas | -10.24 kJ/g | solid | -9.178 kJ/g molar heat of formation Δ_fH° | gas | -1297 kJ/mol | solid | -1163 kJ/mol specific entropy S° | solid | 0.6629 J/(g K) molar entropy S° | solid | 84 J/(mol K) (at STP)
Chemical identifiers
F InChI identifier | InChI=1/3FH.Ga/h3*1H;/q;;;+3/p-3/f3F.Ga/h3*1h;/q3*-1;m MDL number | MFCD00011021](../image_source/0bbf2a3851277559122ba8c6ff1e9b47.png)
CAS number | 7783-51-9 PubChem CID number | 82211 PubChem SID number | 24868640 SMILES identifier | F[Ga](F)F InChI identifier | InChI=1/3FH.Ga/h3*1H;/q;;;+3/p-3/f3F.Ga/h3*1h;/q3*-1;m MDL number | MFCD00011021
NFPA label

NFPA label

NFPA health rating | 3 NFPA fire rating | 0 NFPA reactivity rating | 2