Input interpretation
aluminum (chemical element) | argon (chemical element) | sulfur (chemical element) | chlorine (chemical element)
Periodic table location
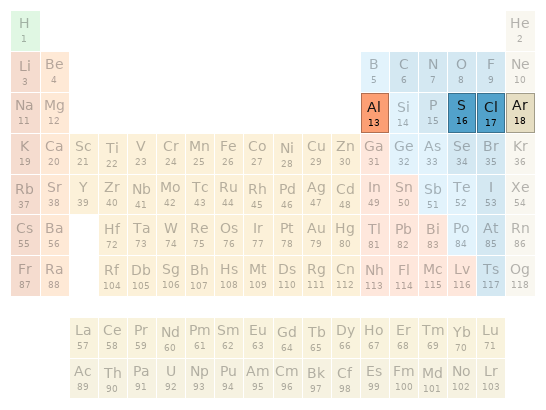
Periodic table location
Images

Images
Basic elemental properties
![| atomic symbol | atomic number | short electronic configuration aluminum | Al | 13 | [Ne]3s^23p^1 argon | Ar | 18 | [Ne]3s^23p^6 sulfur | S | 16 | [Ne]3s^23p^4 chlorine | Cl | 17 | [Ne]3s^23p^5 | Aufbau diagram | block | group aluminum | 3p 3s | p | 13 argon | 3p 3s | p | 18 sulfur | 3p 3s | p | 16 chlorine | 3p 3s | p | 17 | period | atomic mass | half-life aluminum | 3 | 26.9815385 u | (stable) argon | 3 | 39.948 u | (stable) sulfur | 3 | 32.06 u | (stable) chlorine | 3 | 35.45 u | (stable)](../image_source/f7f8c5910de9c8541e35e3cd2be270ba.png)
| atomic symbol | atomic number | short electronic configuration aluminum | Al | 13 | [Ne]3s^23p^1 argon | Ar | 18 | [Ne]3s^23p^6 sulfur | S | 16 | [Ne]3s^23p^4 chlorine | Cl | 17 | [Ne]3s^23p^5 | Aufbau diagram | block | group aluminum | 3p 3s | p | 13 argon | 3p 3s | p | 18 sulfur | 3p 3s | p | 16 chlorine | 3p 3s | p | 17 | period | atomic mass | half-life aluminum | 3 | 26.9815385 u | (stable) argon | 3 | 39.948 u | (stable) sulfur | 3 | 32.06 u | (stable) chlorine | 3 | 35.45 u | (stable)
Thermodynamic properties
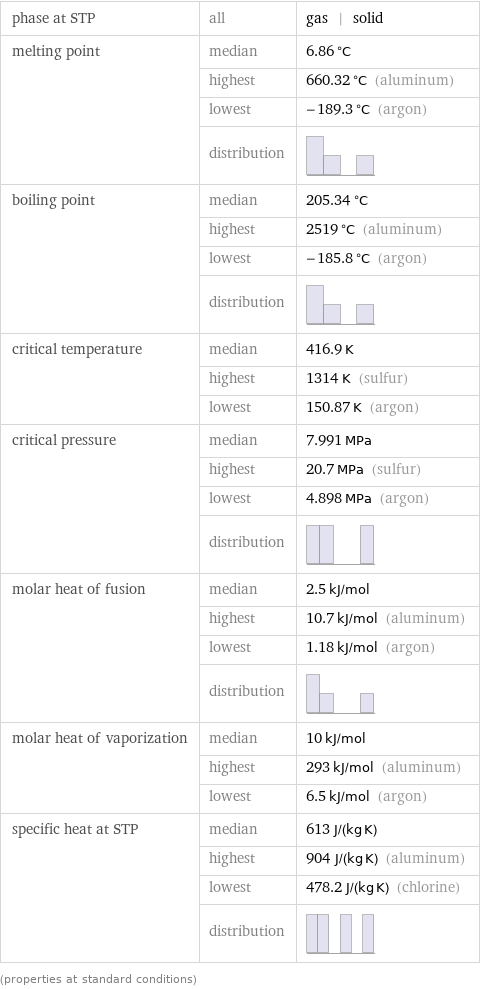
phase at STP | all | gas | solid melting point | median | 6.86 °C | highest | 660.32 °C (aluminum) | lowest | -189.3 °C (argon) | distribution | boiling point | median | 205.34 °C | highest | 2519 °C (aluminum) | lowest | -185.8 °C (argon) | distribution | critical temperature | median | 416.9 K | highest | 1314 K (sulfur) | lowest | 150.87 K (argon) critical pressure | median | 7.991 MPa | highest | 20.7 MPa (sulfur) | lowest | 4.898 MPa (argon) | distribution | molar heat of fusion | median | 2.5 kJ/mol | highest | 10.7 kJ/mol (aluminum) | lowest | 1.18 kJ/mol (argon) | distribution | molar heat of vaporization | median | 10 kJ/mol | highest | 293 kJ/mol (aluminum) | lowest | 6.5 kJ/mol (argon) specific heat at STP | median | 613 J/(kg K) | highest | 904 J/(kg K) (aluminum) | lowest | 478.2 J/(kg K) (chlorine) | distribution | (properties at standard conditions)
Material properties
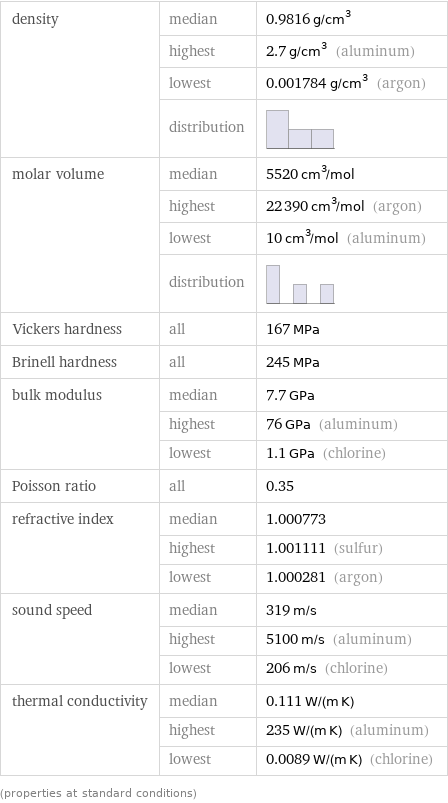
density | median | 0.9816 g/cm^3 | highest | 2.7 g/cm^3 (aluminum) | lowest | 0.001784 g/cm^3 (argon) | distribution | molar volume | median | 5520 cm^3/mol | highest | 22390 cm^3/mol (argon) | lowest | 10 cm^3/mol (aluminum) | distribution | Vickers hardness | all | 167 MPa Brinell hardness | all | 245 MPa bulk modulus | median | 7.7 GPa | highest | 76 GPa (aluminum) | lowest | 1.1 GPa (chlorine) Poisson ratio | all | 0.35 refractive index | median | 1.000773 | highest | 1.001111 (sulfur) | lowest | 1.000281 (argon) sound speed | median | 319 m/s | highest | 5100 m/s (aluminum) | lowest | 206 m/s (chlorine) thermal conductivity | median | 0.111 W/(m K) | highest | 235 W/(m K) (aluminum) | lowest | 0.0089 W/(m K) (chlorine) (properties at standard conditions)
Electromagnetic properties

resistivity | median | 100 Ω m (ohm meters) | highest | 1×10^15 Ω m (ohm meters) (sulfur) | lowest | 2.6×10^-8 Ω m (ohm meters) (aluminum) electrical conductivity | median | 0.01 S/m (siemens per meter) | highest | 3.8×10^7 S/m (siemens per meter) (aluminum) | lowest | 1×10^-15 S/m (siemens per meter) (sulfur) volume magnetic susceptibility | median | -1.69×10^-8 | highest | 2.11×10^-5 (aluminum) | lowest | -1.22×10^-5 (sulfur) | distribution | mass magnetic susceptibility | median | -6.1×10^-9 m^3/kg (cubic meters per kilogram) | highest | 7.8×10^-9 m^3/kg (cubic meters per kilogram) (aluminum) | lowest | -7.2×10^-9 m^3/kg (cubic meters per kilogram) (chlorine) molar magnetic susceptibility | median | -2.2×10^-10 m^3/mol (cubic meters per mole) | highest | 2.1×10^-10 m^3/mol (cubic meters per mole) (aluminum) | lowest | -5.11×10^-10 m^3/mol (cubic meters per mole) (chlorine) | distribution | work function | all | (4.06 to 4.26) eV
Reactivity

valence | median | 4 | highest | 6 (sulfur) | lowest | 0 (argon) | distribution | electronegativity | median | 2.58 | highest | 3.16 (chlorine) | lowest | 1.61 (aluminum) | distribution | electron affinity | median | 120 kJ/mol (kilojoules per mole) | highest | 349 kJ/mol (kilojoules per mole) (chlorine) | lowest | 0 kJ/mol (kilojoules per mole) (argon) | distribution | first ionization energy | median | 1125 kJ/mol (kilojoules per mole) | highest | 1520.6 kJ/mol (kilojoules per mole) (argon) | lowest | 577.5 kJ/mol (kilojoules per mole) (aluminum) | distribution |
Atomic properties
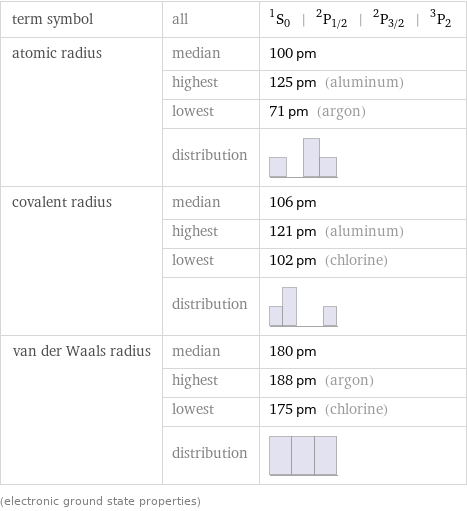
term symbol | all | ^1S_0 | ^2P_(1/2) | ^2P_(3/2) | ^3P_2 atomic radius | median | 100 pm | highest | 125 pm (aluminum) | lowest | 71 pm (argon) | distribution | covalent radius | median | 106 pm | highest | 121 pm (aluminum) | lowest | 102 pm (chlorine) | distribution | van der Waals radius | median | 180 pm | highest | 188 pm (argon) | lowest | 175 pm (chlorine) | distribution | (electronic ground state properties)
Abundances
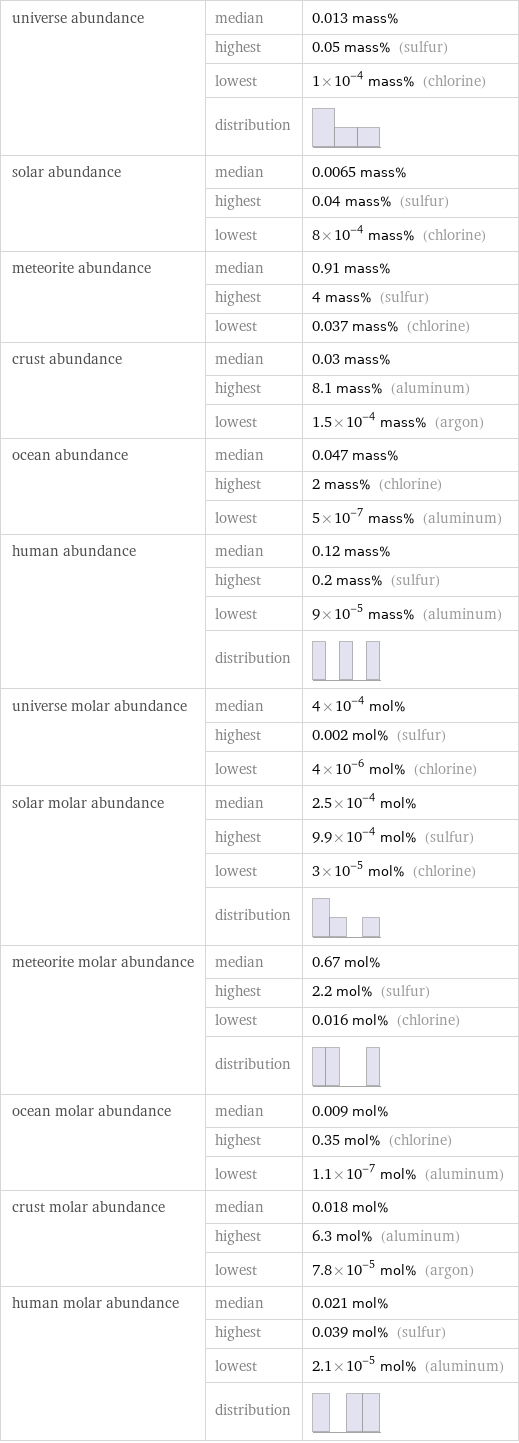
universe abundance | median | 0.013 mass% | highest | 0.05 mass% (sulfur) | lowest | 1×10^-4 mass% (chlorine) | distribution | solar abundance | median | 0.0065 mass% | highest | 0.04 mass% (sulfur) | lowest | 8×10^-4 mass% (chlorine) meteorite abundance | median | 0.91 mass% | highest | 4 mass% (sulfur) | lowest | 0.037 mass% (chlorine) crust abundance | median | 0.03 mass% | highest | 8.1 mass% (aluminum) | lowest | 1.5×10^-4 mass% (argon) ocean abundance | median | 0.047 mass% | highest | 2 mass% (chlorine) | lowest | 5×10^-7 mass% (aluminum) human abundance | median | 0.12 mass% | highest | 0.2 mass% (sulfur) | lowest | 9×10^-5 mass% (aluminum) | distribution | universe molar abundance | median | 4×10^-4 mol% | highest | 0.002 mol% (sulfur) | lowest | 4×10^-6 mol% (chlorine) solar molar abundance | median | 2.5×10^-4 mol% | highest | 9.9×10^-4 mol% (sulfur) | lowest | 3×10^-5 mol% (chlorine) | distribution | meteorite molar abundance | median | 0.67 mol% | highest | 2.2 mol% (sulfur) | lowest | 0.016 mol% (chlorine) | distribution | ocean molar abundance | median | 0.009 mol% | highest | 0.35 mol% (chlorine) | lowest | 1.1×10^-7 mol% (aluminum) crust molar abundance | median | 0.018 mol% | highest | 6.3 mol% (aluminum) | lowest | 7.8×10^-5 mol% (argon) human molar abundance | median | 0.021 mol% | highest | 0.039 mol% (sulfur) | lowest | 2.1×10^-5 mol% (aluminum) | distribution |