Input interpretation

actinides | atomic diameter
Summary

median | 350 pm highest | 390 pm (actinium) lowest | 350 pm (4 elements) distribution | | (based on 7 values; 8 unavailable)
Entities with missing values

curium | berkelium | californium | einsteinium | fermium | mendelevium | nobelium | lawrencium (total: 8)
Plot
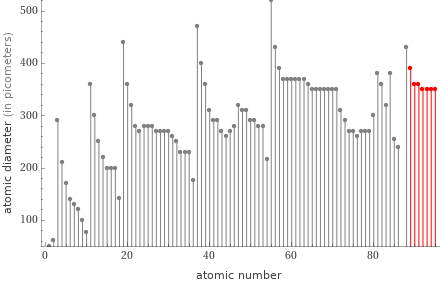
Plot
Periodic table location

Periodic table location
Atomic radii properties
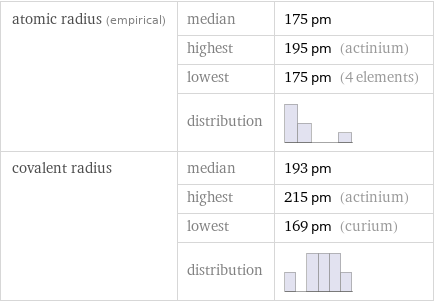
atomic radius (empirical) | median | 175 pm | highest | 195 pm (actinium) | lowest | 175 pm (4 elements) | distribution | covalent radius | median | 193 pm | highest | 215 pm (actinium) | lowest | 169 pm (curium) | distribution |
Units

Atomic diameter rankings
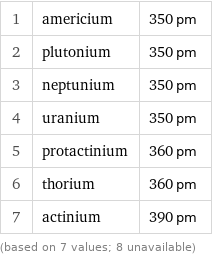
1 | americium | 350 pm 2 | plutonium | 350 pm 3 | neptunium | 350 pm 4 | uranium | 350 pm 5 | protactinium | 360 pm 6 | thorium | 360 pm 7 | actinium | 390 pm (based on 7 values; 8 unavailable)
Unit conversions for median atomic diameter 350 pm
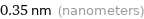
0.35 nm (nanometers)
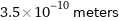
3.5×10^-10 meters
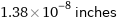
1.38×10^-8 inches
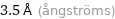
3.5 Å (ångströms)
Comparisons for median atomic diameter 350 pm

≈ 0.4 × carbon nanotube diameter ( ≈ 1 nm )
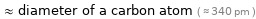
≈ diameter of a carbon atom ( ≈ 340 pm )
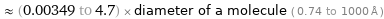
≈ (0.00349 to 4.7) × diameter of a molecule ( 0.74 to 1000 Å )