Input interpretation
sodium bromate | lime
Chemical names and formulas
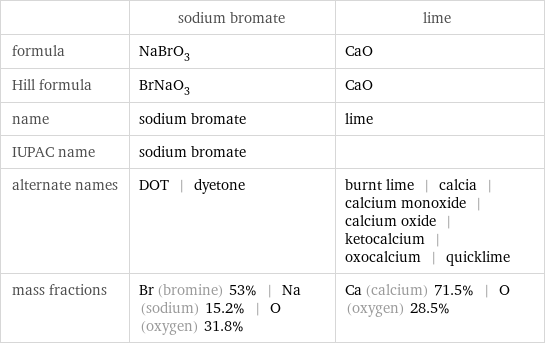
| sodium bromate | lime formula | NaBrO_3 | CaO Hill formula | BrNaO_3 | CaO name | sodium bromate | lime IUPAC name | sodium bromate | alternate names | DOT | dyetone | burnt lime | calcia | calcium monoxide | calcium oxide | ketocalcium | oxocalcium | quicklime mass fractions | Br (bromine) 53% | Na (sodium) 15.2% | O (oxygen) 31.8% | Ca (calcium) 71.5% | O (oxygen) 28.5%
Structure diagrams
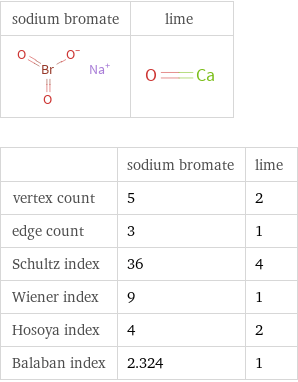
| sodium bromate | lime vertex count | 5 | 2 edge count | 3 | 1 Schultz index | 36 | 4 Wiener index | 9 | 1 Hosoya index | 4 | 2 Balaban index | 2.324 | 1
Basic properties
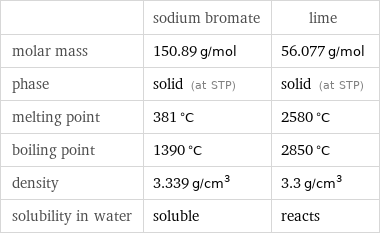
| sodium bromate | lime molar mass | 150.89 g/mol | 56.077 g/mol phase | solid (at STP) | solid (at STP) melting point | 381 °C | 2580 °C boiling point | 1390 °C | 2850 °C density | 3.339 g/cm^3 | 3.3 g/cm^3 solubility in water | soluble | reacts
Units

Thermodynamic properties
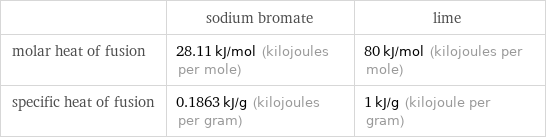
| sodium bromate | lime molar heat of fusion | 28.11 kJ/mol (kilojoules per mole) | 80 kJ/mol (kilojoules per mole) specific heat of fusion | 0.1863 kJ/g (kilojoules per gram) | 1 kJ/g (kilojoule per gram)
Solid properties (at STP)

| sodium bromate | lime density | 3.339 g/cm^3 | 3.3 g/cm^3 vapor pressure | | 1.8×10^-6 mmHg
Units
