Input interpretation
magnesium hydroxide
Chemical names and formulas
![formula | Mg(OH)_2 Hill formula | H_2MgO_2 name | magnesium hydroxide IUPAC name | magnesium dihydroxide alternate names | magnesia magma | magnesia, [Milk of] | magnesium dihydroxide | magnesium hydrate | milk of magnesia mass fractions | H (hydrogen) 3.46% | Mg (magnesium) 41.7% | O (oxygen) 54.9%](../image_source/45a8c837a94dc57628b583159a472622.png)
formula | Mg(OH)_2 Hill formula | H_2MgO_2 name | magnesium hydroxide IUPAC name | magnesium dihydroxide alternate names | magnesia magma | magnesia, [Milk of] | magnesium dihydroxide | magnesium hydrate | milk of magnesia mass fractions | H (hydrogen) 3.46% | Mg (magnesium) 41.7% | O (oxygen) 54.9%
Structure diagram
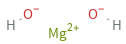
Structure diagram
Basic properties
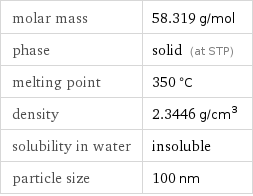
molar mass | 58.319 g/mol phase | solid (at STP) melting point | 350 °C density | 2.3446 g/cm^3 solubility in water | insoluble particle size | 100 nm
Solid properties (at STP)

density | 2.3446 g/cm^3
Units

Thermodynamic properties
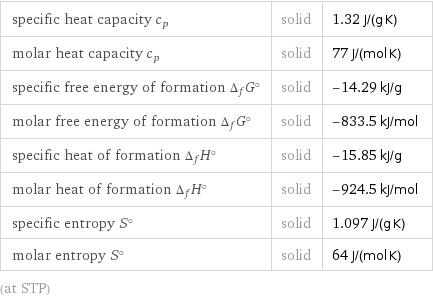
specific heat capacity c_p | solid | 1.32 J/(g K) molar heat capacity c_p | solid | 77 J/(mol K) specific free energy of formation Δ_fG° | solid | -14.29 kJ/g molar free energy of formation Δ_fG° | solid | -833.5 kJ/mol specific heat of formation Δ_fH° | solid | -15.85 kJ/g molar heat of formation Δ_fH° | solid | -924.5 kJ/mol specific entropy S° | solid | 1.097 J/(g K) molar entropy S° | solid | 64 J/(mol K) (at STP)
Chemical identifiers
![CAS number | 1309-42-8 PubChem CID number | 14791 PubChem SID number | 24882320 SMILES identifier | [OH-].[OH-].[Mg+2] InChI identifier | InChI=1/Mg.2H2O/h;2*1H2/q+2;;/p-2/fMg.2HO/h;2*1h/qm;2*-1 RTECS number | OM3570000 MDL number | MFCD00011104](../image_source/1e35b6e94e6126c143f9c376da57c851.png)
CAS number | 1309-42-8 PubChem CID number | 14791 PubChem SID number | 24882320 SMILES identifier | [OH-].[OH-].[Mg+2] InChI identifier | InChI=1/Mg.2H2O/h;2*1H2/q+2;;/p-2/fMg.2HO/h;2*1h/qm;2*-1 RTECS number | OM3570000 MDL number | MFCD00011104
NFPA label

NFPA label

NFPA health rating | 1 NFPA fire rating | 0 NFPA reactivity rating | 0
Toxicity properties

RTECS classes | drug | human data
Ion equivalents

Mg^(2+) (magnesium cation) | 1 (OH)^- (hydroxide anion) | 2