Input interpretation
barium (chemical element) | bismuth (chemical element) | tantalum (chemical element) | europium (chemical element)
Periodic table location
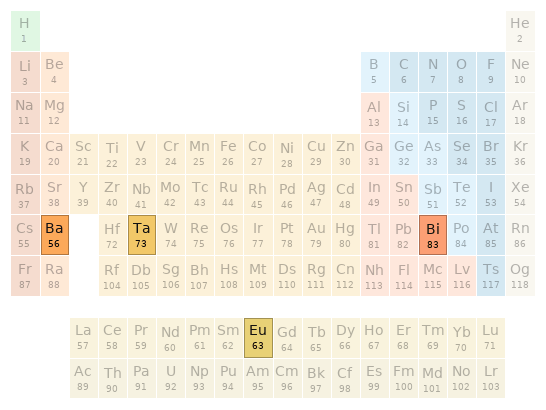
Periodic table location
Images

Images
Basic elemental properties
![| atomic symbol | atomic number | short electronic configuration barium | Ba | 56 | [Xe]6s^2 bismuth | Bi | 83 | [Xe]6s^24f^145d^106p^3 tantalum | Ta | 73 | [Xe]6s^24f^145d^3 europium | Eu | 63 | [Xe]6s^24f^7 | Aufbau diagram | block | group barium | 6s | s | 2 bismuth | 6p 5d 4f 6s | p | 15 tantalum | 5d 4f 6s | d | 5 europium | 4f 6s | f | | period | atomic mass | half-life barium | 6 | 137.327 u | (stable) bismuth | 6 | 208.9804 u | 1.9×10^19 yr tantalum | 6 | 180.94788 u | (stable) europium | 6 | 151.964 u | (stable)](../image_source/78586c8e3f2c76ab88ab9ccad942f215.png)
| atomic symbol | atomic number | short electronic configuration barium | Ba | 56 | [Xe]6s^2 bismuth | Bi | 83 | [Xe]6s^24f^145d^106p^3 tantalum | Ta | 73 | [Xe]6s^24f^145d^3 europium | Eu | 63 | [Xe]6s^24f^7 | Aufbau diagram | block | group barium | 6s | s | 2 bismuth | 6p 5d 4f 6s | p | 15 tantalum | 5d 4f 6s | d | 5 europium | 4f 6s | f | | period | atomic mass | half-life barium | 6 | 137.327 u | (stable) bismuth | 6 | 208.9804 u | 1.9×10^19 yr tantalum | 6 | 180.94788 u | (stable) europium | 6 | 151.964 u | (stable)
Thermodynamic properties
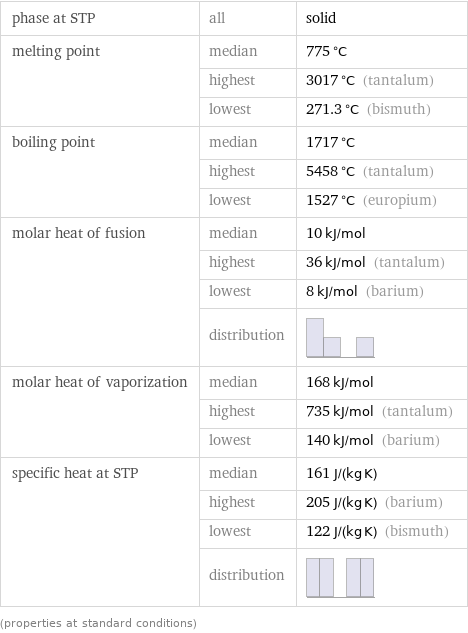
phase at STP | all | solid melting point | median | 775 °C | highest | 3017 °C (tantalum) | lowest | 271.3 °C (bismuth) boiling point | median | 1717 °C | highest | 5458 °C (tantalum) | lowest | 1527 °C (europium) molar heat of fusion | median | 10 kJ/mol | highest | 36 kJ/mol (tantalum) | lowest | 8 kJ/mol (barium) | distribution | molar heat of vaporization | median | 168 kJ/mol | highest | 735 kJ/mol (tantalum) | lowest | 140 kJ/mol (barium) specific heat at STP | median | 161 J/(kg K) | highest | 205 J/(kg K) (barium) | lowest | 122 J/(kg K) (bismuth) | distribution | (properties at standard conditions)
Material properties
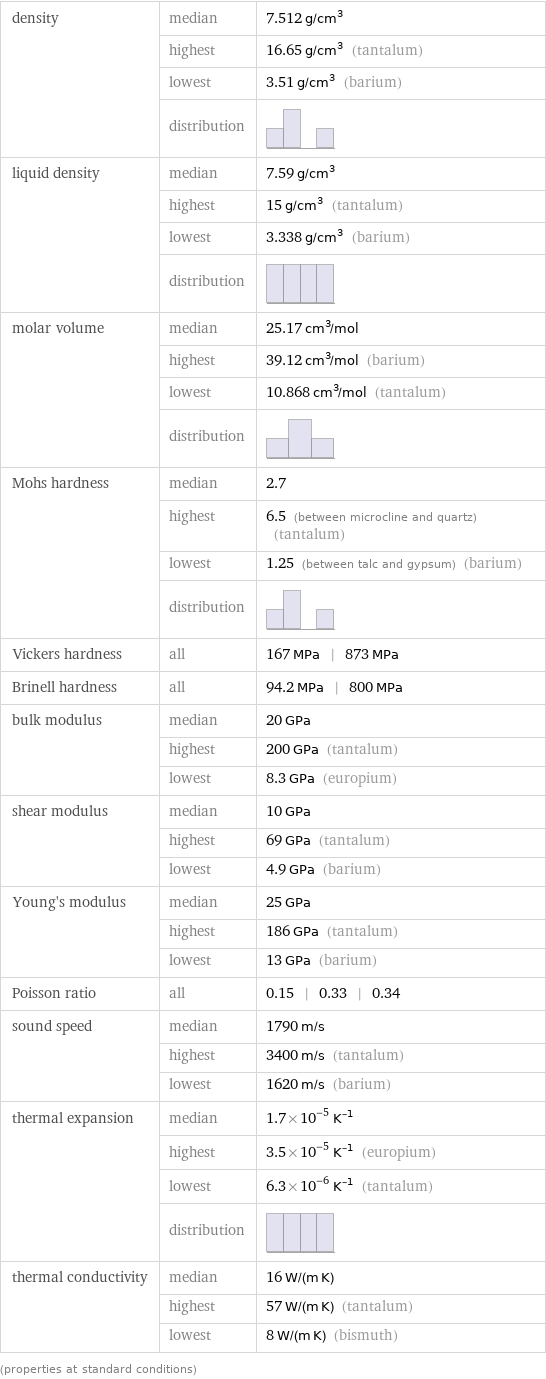
density | median | 7.512 g/cm^3 | highest | 16.65 g/cm^3 (tantalum) | lowest | 3.51 g/cm^3 (barium) | distribution | liquid density | median | 7.59 g/cm^3 | highest | 15 g/cm^3 (tantalum) | lowest | 3.338 g/cm^3 (barium) | distribution | molar volume | median | 25.17 cm^3/mol | highest | 39.12 cm^3/mol (barium) | lowest | 10.868 cm^3/mol (tantalum) | distribution | Mohs hardness | median | 2.7 | highest | 6.5 (between microcline and quartz) (tantalum) | lowest | 1.25 (between talc and gypsum) (barium) | distribution | Vickers hardness | all | 167 MPa | 873 MPa Brinell hardness | all | 94.2 MPa | 800 MPa bulk modulus | median | 20 GPa | highest | 200 GPa (tantalum) | lowest | 8.3 GPa (europium) shear modulus | median | 10 GPa | highest | 69 GPa (tantalum) | lowest | 4.9 GPa (barium) Young's modulus | median | 25 GPa | highest | 186 GPa (tantalum) | lowest | 13 GPa (barium) Poisson ratio | all | 0.15 | 0.33 | 0.34 sound speed | median | 1790 m/s | highest | 3400 m/s (tantalum) | lowest | 1620 m/s (barium) thermal expansion | median | 1.7×10^-5 K^(-1) | highest | 3.5×10^-5 K^(-1) (europium) | lowest | 6.3×10^-6 K^(-1) (tantalum) | distribution | thermal conductivity | median | 16 W/(m K) | highest | 57 W/(m K) (tantalum) | lowest | 8 W/(m K) (bismuth) (properties at standard conditions)
Electromagnetic properties

resistivity | median | 6.3×10^-7 Ω m (ohm meters) | highest | 1.3×10^-6 Ω m (ohm meters) (bismuth) | lowest | 1.3×10^-7 Ω m (ohm meters) (tantalum) | distribution | electrical conductivity | median | 2×10^6 S/m (siemens per meter) | highest | 7.7×10^6 S/m (siemens per meter) (tantalum) | lowest | 770000 S/m (siemens per meter) (bismuth) | distribution | volume magnetic susceptibility | median | 1.089×10^-4 | highest | 0.0014473 (europium) | lowest | -1.7×10^-4 (bismuth) | distribution | mass magnetic susceptibility | median | 1.1×10^-8 m^3/kg (cubic meters per kilogram) | highest | 2.76×10^-7 m^3/kg (cubic meters per kilogram) (europium) | lowest | -1.7×10^-8 m^3/kg (cubic meters per kilogram) (bismuth) | distribution | molar magnetic susceptibility | median | 1.744×10^-9 m^3/mol (cubic meters per mole) | highest | 4.1942×10^-8 m^3/mol (cubic meters per mole) (europium) | lowest | -3.6×10^-9 m^3/mol (cubic meters per mole) (bismuth) | distribution | work function | all | 2.5 eV | 2.52 eV | 4.34 eV | (4 to 4.8) eV
Reactivity
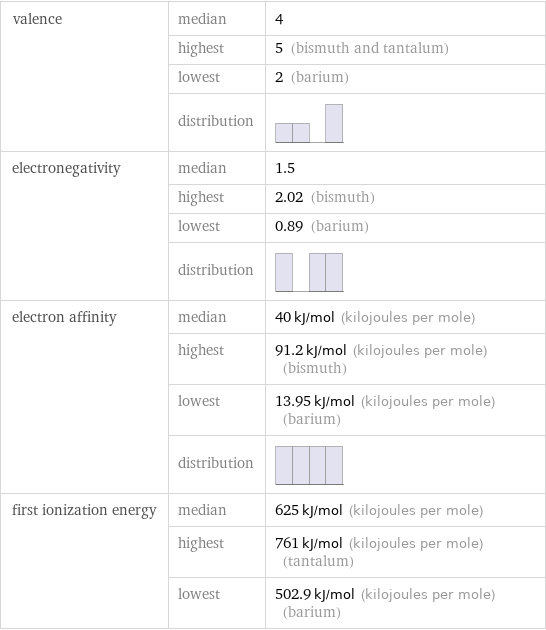
valence | median | 4 | highest | 5 (bismuth and tantalum) | lowest | 2 (barium) | distribution | electronegativity | median | 1.5 | highest | 2.02 (bismuth) | lowest | 0.89 (barium) | distribution | electron affinity | median | 40 kJ/mol (kilojoules per mole) | highest | 91.2 kJ/mol (kilojoules per mole) (bismuth) | lowest | 13.95 kJ/mol (kilojoules per mole) (barium) | distribution | first ionization energy | median | 625 kJ/mol (kilojoules per mole) | highest | 761 kJ/mol (kilojoules per mole) (tantalum) | lowest | 502.9 kJ/mol (kilojoules per mole) (barium)
Atomic properties
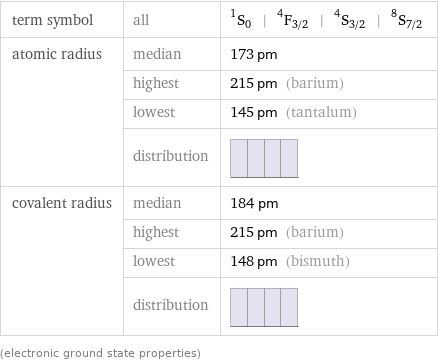
term symbol | all | ^1S_0 | ^4F_(3/2) | ^4S_(3/2) | ^8S_(7/2) atomic radius | median | 173 pm | highest | 215 pm (barium) | lowest | 145 pm (tantalum) | distribution | covalent radius | median | 184 pm | highest | 215 pm (barium) | lowest | 148 pm (bismuth) | distribution | (electronic ground state properties)
Abundances
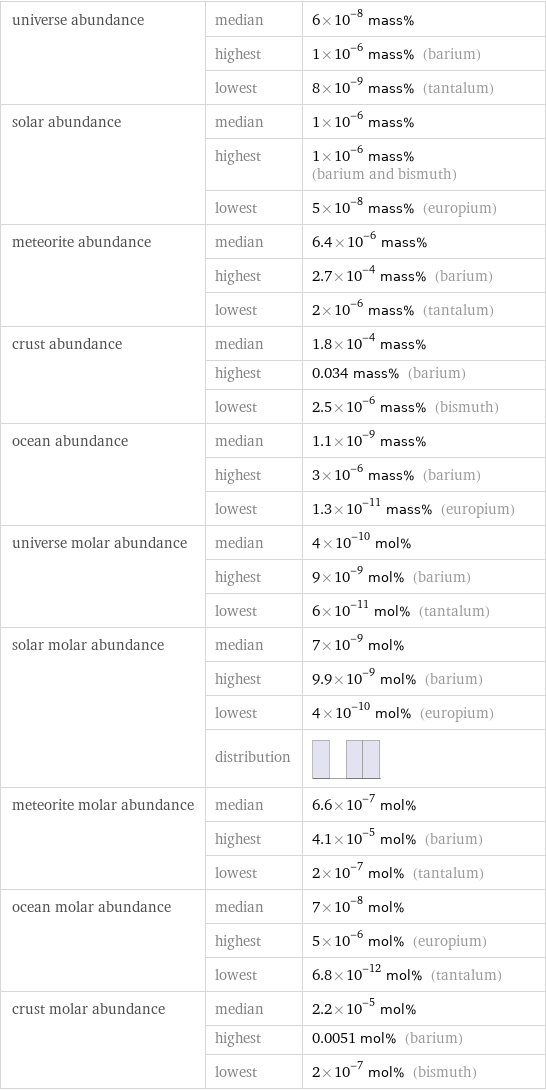
universe abundance | median | 6×10^-8 mass% | highest | 1×10^-6 mass% (barium) | lowest | 8×10^-9 mass% (tantalum) solar abundance | median | 1×10^-6 mass% | highest | 1×10^-6 mass% (barium and bismuth) | lowest | 5×10^-8 mass% (europium) meteorite abundance | median | 6.4×10^-6 mass% | highest | 2.7×10^-4 mass% (barium) | lowest | 2×10^-6 mass% (tantalum) crust abundance | median | 1.8×10^-4 mass% | highest | 0.034 mass% (barium) | lowest | 2.5×10^-6 mass% (bismuth) ocean abundance | median | 1.1×10^-9 mass% | highest | 3×10^-6 mass% (barium) | lowest | 1.3×10^-11 mass% (europium) universe molar abundance | median | 4×10^-10 mol% | highest | 9×10^-9 mol% (barium) | lowest | 6×10^-11 mol% (tantalum) solar molar abundance | median | 7×10^-9 mol% | highest | 9.9×10^-9 mol% (barium) | lowest | 4×10^-10 mol% (europium) | distribution | meteorite molar abundance | median | 6.6×10^-7 mol% | highest | 4.1×10^-5 mol% (barium) | lowest | 2×10^-7 mol% (tantalum) ocean molar abundance | median | 7×10^-8 mol% | highest | 5×10^-6 mol% (europium) | lowest | 6.8×10^-12 mol% (tantalum) crust molar abundance | median | 2.2×10^-5 mol% | highest | 0.0051 mol% (barium) | lowest | 2×10^-7 mol% (bismuth)