Input interpretation
carbon (chemical element) | hydrogen (chemical element) | antimony (chemical element) | argon (chemical element)
Periodic table location
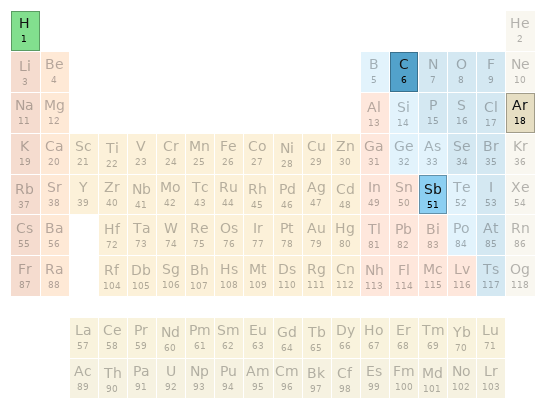
Periodic table location
Images

Images
Basic elemental properties
![| atomic symbol | atomic number | short electronic configuration carbon | C | 6 | [He]2s^22p^2 hydrogen | H | 1 | 1s^1 antimony | Sb | 51 | [Kr]5s^24d^105p^3 argon | Ar | 18 | [Ne]3s^23p^6 | Aufbau diagram | block | group carbon | 2p 2s | p | 14 hydrogen | 1s | s | 1 antimony | 5p 4d 5s | p | 15 argon | 3p 3s | p | 18 | period | atomic mass | half-life carbon | 2 | 12.011 u | (stable) hydrogen | 1 | 1.008 u | (stable) antimony | 5 | 121.76 u | (stable) argon | 3 | 39.948 u | (stable)](../image_source/0ec451c69eb88b95d44f3f479b7f3f3b.png)
| atomic symbol | atomic number | short electronic configuration carbon | C | 6 | [He]2s^22p^2 hydrogen | H | 1 | 1s^1 antimony | Sb | 51 | [Kr]5s^24d^105p^3 argon | Ar | 18 | [Ne]3s^23p^6 | Aufbau diagram | block | group carbon | 2p 2s | p | 14 hydrogen | 1s | s | 1 antimony | 5p 4d 5s | p | 15 argon | 3p 3s | p | 18 | period | atomic mass | half-life carbon | 2 | 12.011 u | (stable) hydrogen | 1 | 1.008 u | (stable) antimony | 5 | 121.76 u | (stable) argon | 3 | 39.948 u | (stable)
Thermodynamic properties
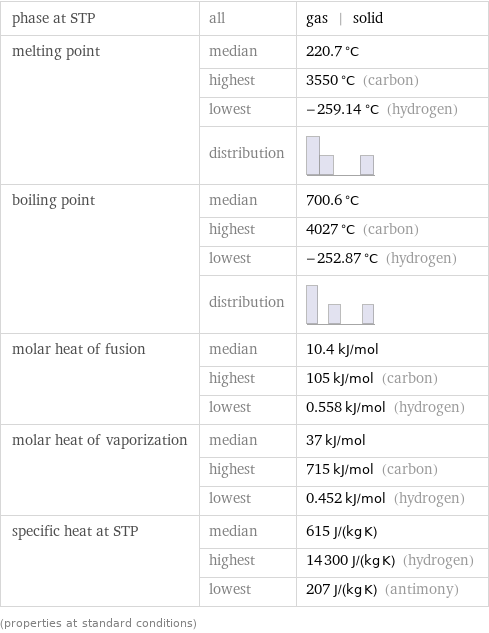
phase at STP | all | gas | solid melting point | median | 220.7 °C | highest | 3550 °C (carbon) | lowest | -259.14 °C (hydrogen) | distribution | boiling point | median | 700.6 °C | highest | 4027 °C (carbon) | lowest | -252.87 °C (hydrogen) | distribution | molar heat of fusion | median | 10.4 kJ/mol | highest | 105 kJ/mol (carbon) | lowest | 0.558 kJ/mol (hydrogen) molar heat of vaporization | median | 37 kJ/mol | highest | 715 kJ/mol (carbon) | lowest | 0.452 kJ/mol (hydrogen) specific heat at STP | median | 615 J/(kg K) | highest | 14300 J/(kg K) (hydrogen) | lowest | 207 J/(kg K) (antimony) (properties at standard conditions)
Material properties
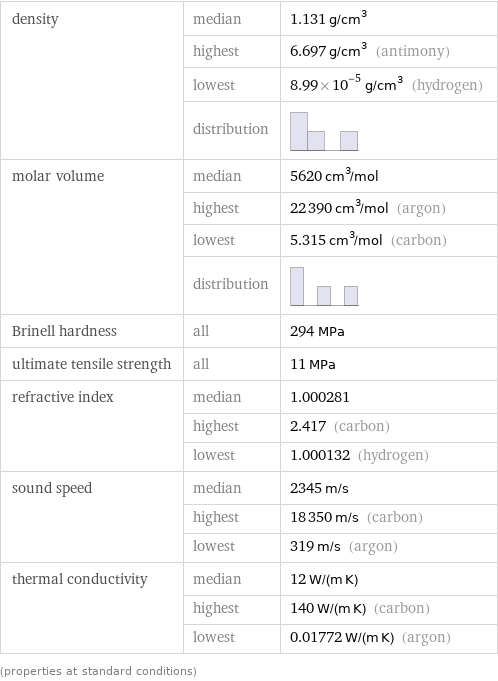
density | median | 1.131 g/cm^3 | highest | 6.697 g/cm^3 (antimony) | lowest | 8.99×10^-5 g/cm^3 (hydrogen) | distribution | molar volume | median | 5620 cm^3/mol | highest | 22390 cm^3/mol (argon) | lowest | 5.315 cm^3/mol (carbon) | distribution | Brinell hardness | all | 294 MPa ultimate tensile strength | all | 11 MPa refractive index | median | 1.000281 | highest | 2.417 (carbon) | lowest | 1.000132 (hydrogen) sound speed | median | 2345 m/s | highest | 18350 m/s (carbon) | lowest | 319 m/s (argon) thermal conductivity | median | 12 W/(m K) | highest | 140 W/(m K) (carbon) | lowest | 0.01772 W/(m K) (argon) (properties at standard conditions)
Reactivity

valence | median | 2.5 | highest | 5 (antimony) | lowest | 0 (argon) electronegativity | median | 2.2 | highest | 2.55 (carbon) | lowest | 2.05 (antimony) | distribution | electron affinity | median | 88 kJ/mol (kilojoules per mole) | highest | 153.9 kJ/mol (kilojoules per mole) (carbon) | lowest | 0 kJ/mol (kilojoules per mole) (argon) | distribution | first ionization energy | median | 1199.3 kJ/mol (kilojoules per mole) | highest | 1520.6 kJ/mol (kilojoules per mole) (argon) | lowest | 834 kJ/mol (kilojoules per mole) (antimony) | distribution |
Atomic properties
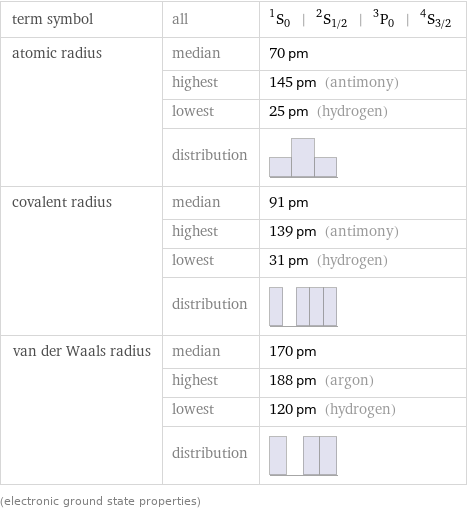
term symbol | all | ^1S_0 | ^2S_(1/2) | ^3P_0 | ^4S_(3/2) atomic radius | median | 70 pm | highest | 145 pm (antimony) | lowest | 25 pm (hydrogen) | distribution | covalent radius | median | 91 pm | highest | 139 pm (antimony) | lowest | 31 pm (hydrogen) | distribution | van der Waals radius | median | 170 pm | highest | 188 pm (argon) | lowest | 120 pm (hydrogen) | distribution | (electronic ground state properties)
Abundances
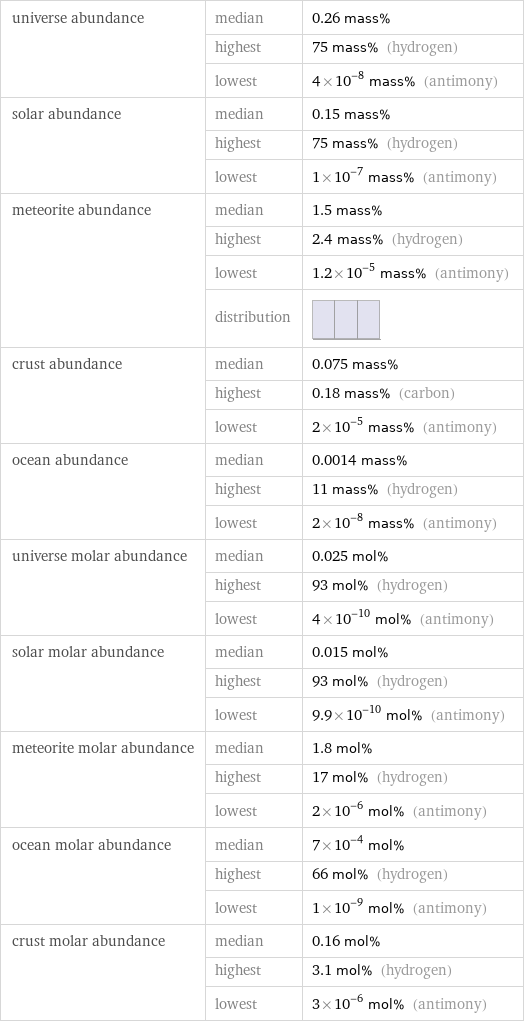
universe abundance | median | 0.26 mass% | highest | 75 mass% (hydrogen) | lowest | 4×10^-8 mass% (antimony) solar abundance | median | 0.15 mass% | highest | 75 mass% (hydrogen) | lowest | 1×10^-7 mass% (antimony) meteorite abundance | median | 1.5 mass% | highest | 2.4 mass% (hydrogen) | lowest | 1.2×10^-5 mass% (antimony) | distribution | crust abundance | median | 0.075 mass% | highest | 0.18 mass% (carbon) | lowest | 2×10^-5 mass% (antimony) ocean abundance | median | 0.0014 mass% | highest | 11 mass% (hydrogen) | lowest | 2×10^-8 mass% (antimony) universe molar abundance | median | 0.025 mol% | highest | 93 mol% (hydrogen) | lowest | 4×10^-10 mol% (antimony) solar molar abundance | median | 0.015 mol% | highest | 93 mol% (hydrogen) | lowest | 9.9×10^-10 mol% (antimony) meteorite molar abundance | median | 1.8 mol% | highest | 17 mol% (hydrogen) | lowest | 2×10^-6 mol% (antimony) ocean molar abundance | median | 7×10^-4 mol% | highest | 66 mol% (hydrogen) | lowest | 1×10^-9 mol% (antimony) crust molar abundance | median | 0.16 mol% | highest | 3.1 mol% (hydrogen) | lowest | 3×10^-6 mol% (antimony)