Input interpretation
ethanol
Chemical names and formulas

formula | CH_3CH_2OH Hill formula | C_2H_6O name | ethanol alternate names | algrain | anhydrol | fermentation alcohol | grain alcohol | potato alcohol | tecsol mass fractions | C (carbon) 52.1% | H (hydrogen) 13.1% | O (oxygen) 34.7%
Lewis structure
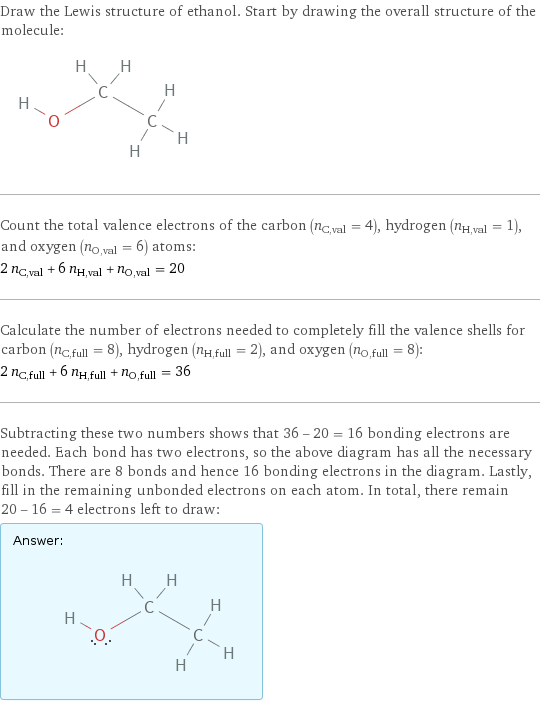
Draw the Lewis structure of ethanol. Start by drawing the overall structure of the molecule: Count the total valence electrons of the carbon (n_C, val = 4), hydrogen (n_H, val = 1), and oxygen (n_O, val = 6) atoms: 2 n_C, val + 6 n_H, val + n_O, val = 20 Calculate the number of electrons needed to completely fill the valence shells for carbon (n_C, full = 8), hydrogen (n_H, full = 2), and oxygen (n_O, full = 8): 2 n_C, full + 6 n_H, full + n_O, full = 36 Subtracting these two numbers shows that 36 - 20 = 16 bonding electrons are needed. Each bond has two electrons, so the above diagram has all the necessary bonds. There are 8 bonds and hence 16 bonding electrons in the diagram. Lastly, fill in the remaining unbonded electrons on each atom. In total, there remain 20 - 16 = 4 electrons left to draw: Answer: | |
3D structure
3D structure
Basic properties
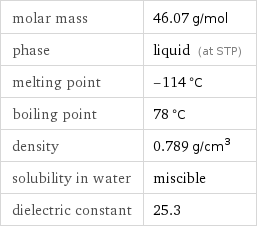
molar mass | 46.07 g/mol phase | liquid (at STP) melting point | -114 °C boiling point | 78 °C density | 0.789 g/cm^3 solubility in water | miscible dielectric constant | 25.3
Liquid properties (at STP)
density | 0.789 g/cm^3 vapor pressure | 44.59 mmHg dynamic viscosity | 0.001074 Pa s (at 25 °C) surface tension | 0.02275 N/m refractive index | 1.36 UV cutoff wavelength | 210 nm
Units

Thermodynamic properties
specific heat capacity c_p | gas | 1.424 J/(g K) | liquid | 2.438 J/(g K) molar heat capacity c_p | gas | 65.6 J/(mol K) | liquid | 112.3 J/(mol K) specific free energy of formation Δ_fG° | gas | -3.645 kJ/g | liquid | -3.794 kJ/g molar free energy of formation Δ_fG° | gas | -167.9 kJ/mol | liquid | -174.8 kJ/mol specific heat of formation Δ_fH° | liquid | -6.028 kJ/g | gas | -5.097 kJ/g molar heat of formation Δ_fH° | liquid | -277.7 kJ/mol | gas | -234.8 kJ/mol specific entropy S° | gas | 6.136 J/(g K) | liquid | 3.488 J/(g K) molar entropy S° | gas | 282.7 J/(mol K) | liquid | 160.7 J/(mol K) molar heat of vaporization | 42.6 kJ/mol | specific heat of vaporization | 0.925 kJ/g | molar heat of combustion | 1368 kJ/mol | specific heat of combustion | 29.69 kJ/g | molar heat of fusion | 4.931 kJ/mol | specific heat of fusion | 0.107 kJ/g | thermal conductivity | 0.1645 W/(m K) | critical temperature | 516 K | critical pressure | 6.4 MPa | (at STP)
Phase diagram
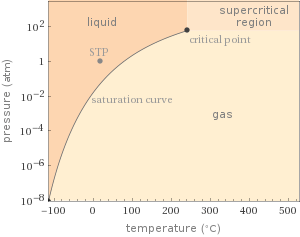
Phase diagram
Units

Basic drug properties

approval status | approved | small molecule drug categories | local anti-infective agent | central nervous system depressant | disinfectant | solvent dosage forms | topical: aerosol | topical: gel | intramuscular: liquid | intravenous: liquid | oral: liquid | topical: liquid | topical: lotion | topical: solution | oral: solution / drops | topical: spray

brand names | aethanol | aethylalkohol | alcare hand degermer | algrain | anhydrol | colamine | denatured alcohol Cd-10 | denatured alcohol Cd-5 | denatured alcohol Cd-5a | denatured alcohol Sd-1 | denatured alcohol Sd-13a | denatured alcohol Sd-17 | denatured alcohol Sd-23a | denatured alcohol Sd-28 | denatured alcohol Sd-30 | denatured alcohol Sd-39b | denatured alcohol Sd-39c | denatured alcohol Sd-3a | denatured alcohol Sd-40m | envision conditioner pdd 9020 | ethanol absolute | ethanol absolute Bp | ethanol, silent spirit | ethyl alcohol & water, 10% | ethyl alcohol & water, 20% | ethyl alcohol & water, 30% | ethyl alcohol & water, 40% | ethyl alcohol & water, 5% | ethyl alcohol & water, 50% | ethyl alcohol & water, 60% | ethyl alcohol & water, 70% | ethyl alcohol & water, 80% | ethyl alcohol & water, 95% | ethyl alcohol & water, 96% | glycinol | jaysol | jaysol S | methylated spirit mineralised | pyro | reagent alcohol | spirit | synasol | tecsol | tecsol C | thanol | thiofaco M-50 addiction potential of drug | 81.8%
Hydrophobicity and permeability properties

experimental LogP hydrophobicity | -0.1 predicted LogP hydrophobicity | -0.4 predicted LogS | 1.1
Chemical identifiers

CAS number | 64-17-5 Beilstein number | 1718733 PubChem CID number | 702 PubChem SID number | 24851272 SMILES identifier | CCO InChI identifier | InChI=1/C2H6O/c1-2-3/h3H, 2H2, 1H3 InChI key | LFQSCWFLJHTTHZ-UHFFFAOYAB MDL number | MFCD00003568