Input interpretation
iodine bromide | hydrogen chloride
Chemical names and formulas
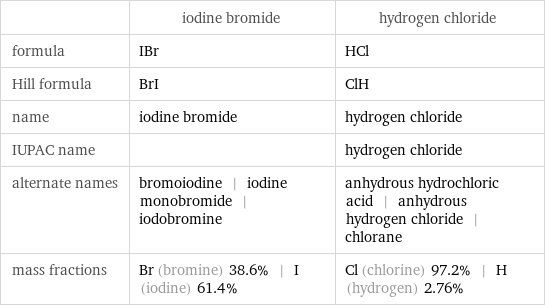
| iodine bromide | hydrogen chloride formula | IBr | HCl Hill formula | BrI | ClH name | iodine bromide | hydrogen chloride IUPAC name | | hydrogen chloride alternate names | bromoiodine | iodine monobromide | iodobromine | anhydrous hydrochloric acid | anhydrous hydrogen chloride | chlorane mass fractions | Br (bromine) 38.6% | I (iodine) 61.4% | Cl (chlorine) 97.2% | H (hydrogen) 2.76%
Structure diagrams

| iodine bromide | hydrogen chloride vertex count | 2 | 1 edge count | 1 | 1 Schultz index | 4 | 0 Wiener index | 1 | 0 Hosoya index | 2 | 1 Balaban index | 1 | 0
3D structure

3D structure
Basic properties
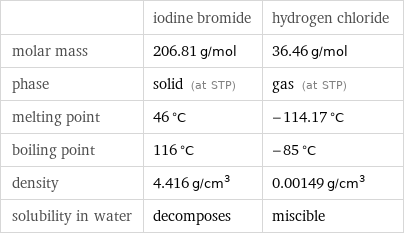
| iodine bromide | hydrogen chloride molar mass | 206.81 g/mol | 36.46 g/mol phase | solid (at STP) | gas (at STP) melting point | 46 °C | -114.17 °C boiling point | 116 °C | -85 °C density | 4.416 g/cm^3 | 0.00149 g/cm^3 solubility in water | decomposes | miscible
Units

Gas properties
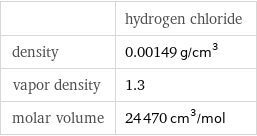
| hydrogen chloride density | 0.00149 g/cm^3 vapor density | 1.3 molar volume | 24470 cm^3/mol
Units

Liquid properties

| hydrogen chloride sound speed | 1066 km/h
Units
