Input interpretation

germanium (chemical element)
Periodic table location
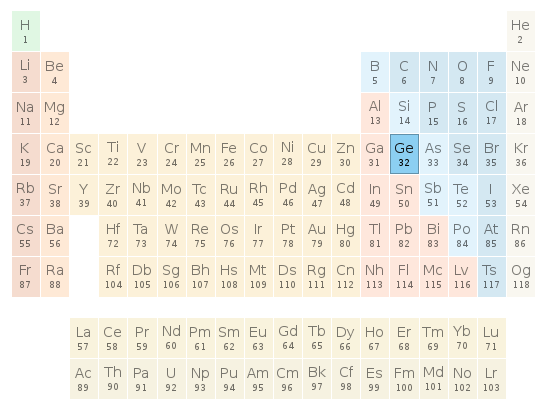
Periodic table location
Image

Image
Basic elemental properties
![atomic symbol | Ge atomic number | 32 short electronic configuration | [Ar]4s^23d^104p^2 Aufbau diagram | 4p 3d 4s block | p group | 14 period | 4 atomic mass | 72.63 u (unified atomic mass units)](../image_source/991247b39f50b97060cc77a4fcb50e4a.png)
atomic symbol | Ge atomic number | 32 short electronic configuration | [Ar]4s^23d^104p^2 Aufbau diagram | 4p 3d 4s block | p group | 14 period | 4 atomic mass | 72.63 u (unified atomic mass units)
Thermodynamic properties
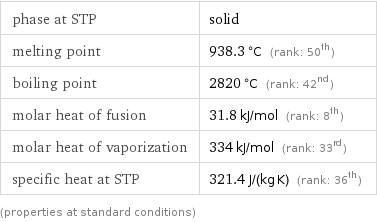
phase at STP | solid melting point | 938.3 °C (rank: 50th) boiling point | 2820 °C (rank: 42nd) molar heat of fusion | 31.8 kJ/mol (rank: 8th) molar heat of vaporization | 334 kJ/mol (rank: 33rd) specific heat at STP | 321.4 J/(kg K) (rank: 36th) (properties at standard conditions)
Material properties
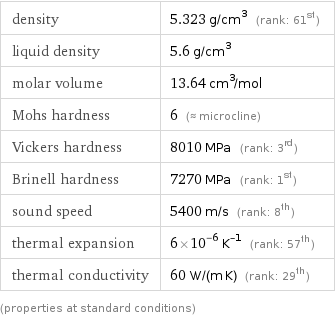
density | 5.323 g/cm^3 (rank: 61st) liquid density | 5.6 g/cm^3 molar volume | 13.64 cm^3/mol Mohs hardness | 6 (≈ microcline) Vickers hardness | 8010 MPa (rank: 3rd) Brinell hardness | 7270 MPa (rank: 1st) sound speed | 5400 m/s (rank: 8th) thermal expansion | 6×10^-6 K^(-1) (rank: 57th) thermal conductivity | 60 W/(m K) (rank: 29th) (properties at standard conditions)
Electromagnetic properties
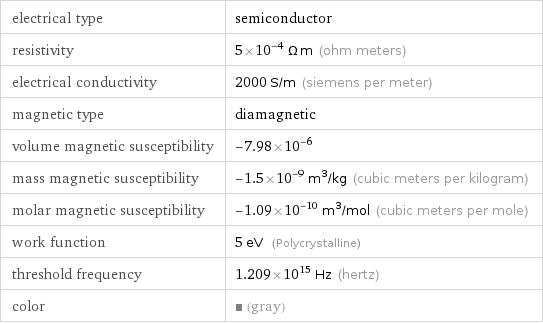
electrical type | semiconductor resistivity | 5×10^-4 Ω m (ohm meters) electrical conductivity | 2000 S/m (siemens per meter) magnetic type | diamagnetic volume magnetic susceptibility | -7.98×10^-6 mass magnetic susceptibility | -1.5×10^-9 m^3/kg (cubic meters per kilogram) molar magnetic susceptibility | -1.09×10^-10 m^3/mol (cubic meters per mole) work function | 5 eV (Polycrystalline) threshold frequency | 1.209×10^15 Hz (hertz) color | (gray)
Reactivity
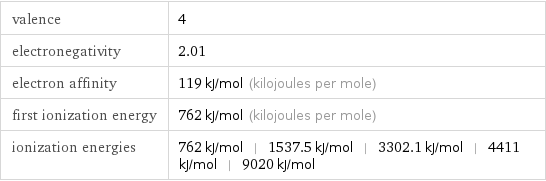
valence | 4 electronegativity | 2.01 electron affinity | 119 kJ/mol (kilojoules per mole) first ionization energy | 762 kJ/mol (kilojoules per mole) ionization energies | 762 kJ/mol | 1537.5 kJ/mol | 3302.1 kJ/mol | 4411 kJ/mol | 9020 kJ/mol
Reactivity
Atomic properties

term symbol | ^3P_0 atomic radius | 125 pm covalent radius | 120 pm (electronic ground state properties)
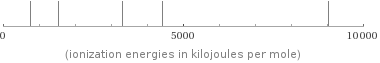
(neutral germanium atomic spectrum)
Abundances
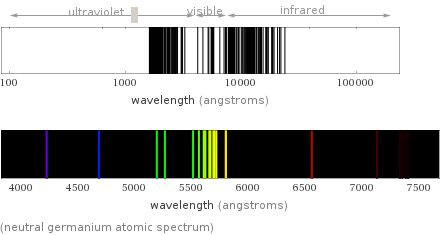
universe abundance | 2×10^-5 mass% (rank: 26th) solar abundance | 2×10^-5 mass% (rank: 27th) meteorite abundance | 0.0021 mass% (rank: 24th) crust abundance | 1.4×10^-4 mass% (rank: 55th) ocean abundance | 6×10^-9 mass% (rank: 42nd) universe molar abundance | 3×10^-7 mol% (rank: 26th) solar molar abundance | 3×10^-7 mol% (rank: 27th) meteorite molar abundance | 4.9×10^-4 mol% (rank: 24th) ocean molar abundance | 5.1×10^-10 mol% (rank: 53rd) crust molar abundance | 3.9×10^-5 mol% (rank: 47th)
Nuclear properties

half-life | (stable) stable isotopes | Ge-74 (36.73%) | Ge-72 (27.31%) | Ge-70 (20.37%) | Ge-73 (7.76%) nuclear spin | Ge-58: 0^+ | Ge-59: 7/2^- | Ge-60: 0^+ | Ge-61: 3/2^- | Ge-62: 0^+ | Ge-63: 3/2^- | Ge-64: 0^+ | Ge-65: 3/2^- | Ge-66: 0^+ | Ge-67: 1/2^- | Ge-68: 0^+ | Ge-69: 5/2^- | Ge-70: 0^+ | Ge-71: 1/2^- | Ge-72: 0^+ | Ge-73: 9/2^+ | Ge-74: 0^+ | Ge-75: 1/2^- | Ge-76: 0^+ | Ge-77: 7/2^+ | Ge-78: 0^+ | Ge-79: 1/2^- | Ge-80: 0^+ | Ge-81: 9/2^+ | Ge-82: 0^+ | Ge-83: 5/2^+ | Ge-84: 0^+ | Ge-85: 5/2^+ | Ge-86: 0^+ | Ge-87: 5/2^+ | Ge-88: 0^+ | Ge-89: 3/2^+ unstable isotopes | Ge-68 (270.9 days) | Ge-71 (11.43 days) | Ge-69 (39.06 h) | Ge-77 (11.3 h) | Ge-66 (135.7 min) | Ge-78 (88 min) | Ge-75 (82.78 min) | Ge-67 (18.9 min) | Ge-64 (63.7 s) | Ge-65 (30.9 s) | Ge-80 (29.5 s) | Ge-79 (18.98 s) | Ge-81 (7.6 s) | Ge-82 (4.55 s) | Ge-83 (1.85 s) | Ge-84 (947 ms) | Ge-85 (535 ms) | Ge-63 (142 ms) | Ge-87 (140 ms) | Ge-89 (50 ms) | Ge-61 (39 ms) | Ge-60 (30 ms) | Ge-88 (300 ns) | Ge-86 (150 ns) | Ge-62 (150 ns) neutron cross-section | 2.2 b neutron mass absorption | 0.0011 m^2/kg
Identifiers

CAS number | 7440-56-4 PubChem CID number | 6326954