Input interpretation
chlorine trifluoride | hydrogen
Chemical names and formulas

| chlorine trifluoride | hydrogen formula | ClF_3 | H_2 Hill formula | ClF_3 | H_2 name | chlorine trifluoride | hydrogen IUPAC name | | molecular hydrogen alternate names | chlorine fluoride | chlorotrifluoride | trifluorure de chlore | dihydrogen | Freon 702 | hydrogen gas | molecular hydrogen | R-702 mass fractions | Cl (chlorine) 38.3% | F (fluorine) 61.7% | H (hydrogen) 100%
Structure diagrams
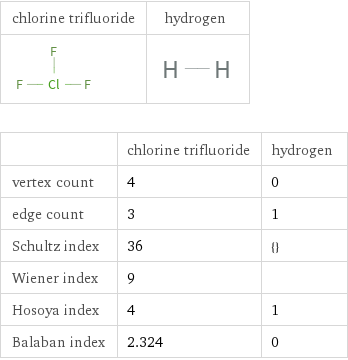
| chlorine trifluoride | hydrogen vertex count | 4 | 0 edge count | 3 | 1 Schultz index | 36 | {} Wiener index | 9 | Hosoya index | 4 | 1 Balaban index | 2.324 | 0
3D structure
3D structure
Basic properties
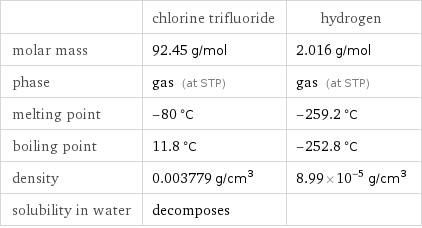
| chlorine trifluoride | hydrogen molar mass | 92.45 g/mol | 2.016 g/mol phase | gas (at STP) | gas (at STP) melting point | -80 °C | -259.2 °C boiling point | 11.8 °C | -252.8 °C density | 0.003779 g/cm^3 | 8.99×10^-5 g/cm^3 solubility in water | decomposes |
Units
