Input interpretation

gold silver | melting point
Results
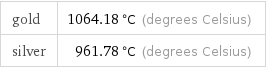
gold | 1064.18 °C (degrees Celsius) silver | 961.78 °C (degrees Celsius)
Plot
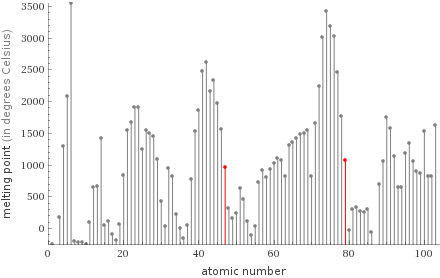
Plot
Thermodynamic properties
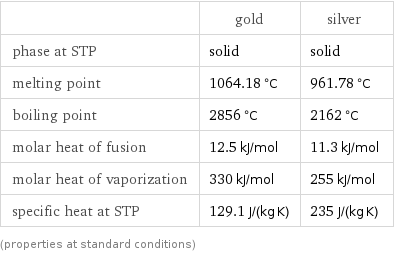
| gold | silver phase at STP | solid | solid melting point | 1064.18 °C | 961.78 °C boiling point | 2856 °C | 2162 °C molar heat of fusion | 12.5 kJ/mol | 11.3 kJ/mol molar heat of vaporization | 330 kJ/mol | 255 kJ/mol specific heat at STP | 129.1 J/(kg K) | 235 J/(kg K) (properties at standard conditions)