Input interpretation
gray antimony | scandium
Chemical names and formulas
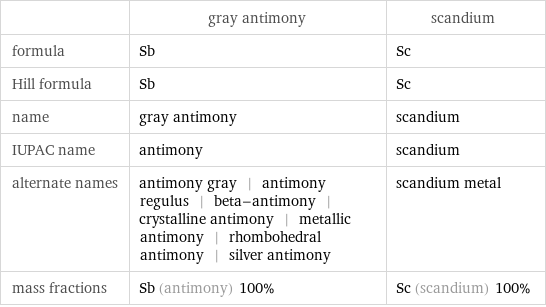
| gray antimony | scandium formula | Sb | Sc Hill formula | Sb | Sc name | gray antimony | scandium IUPAC name | antimony | scandium alternate names | antimony gray | antimony regulus | beta-antimony | crystalline antimony | metallic antimony | rhombohedral antimony | silver antimony | scandium metal mass fractions | Sb (antimony) 100% | Sc (scandium) 100%
Structure diagrams

Structure diagrams
Basic properties
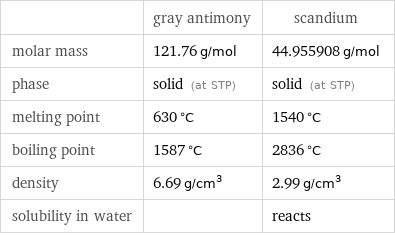
| gray antimony | scandium molar mass | 121.76 g/mol | 44.955908 g/mol phase | solid (at STP) | solid (at STP) melting point | 630 °C | 1540 °C boiling point | 1587 °C | 2836 °C density | 6.69 g/cm^3 | 2.99 g/cm^3 solubility in water | | reacts
Units

Thermodynamic properties
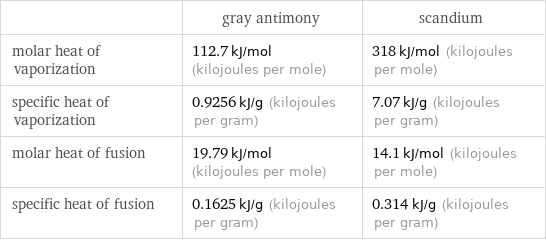
| gray antimony | scandium molar heat of vaporization | 112.7 kJ/mol (kilojoules per mole) | 318 kJ/mol (kilojoules per mole) specific heat of vaporization | 0.9256 kJ/g (kilojoules per gram) | 7.07 kJ/g (kilojoules per gram) molar heat of fusion | 19.79 kJ/mol (kilojoules per mole) | 14.1 kJ/mol (kilojoules per mole) specific heat of fusion | 0.1625 kJ/g (kilojoules per gram) | 0.314 kJ/g (kilojoules per gram)
Solid properties (at STP)

| gray antimony | scandium density | 6.69 g/cm^3 | 2.99 g/cm^3
Units

Chemical identifiers
![| gray antimony | scandium CAS number | 7440-36-0 | 7440-20-2 PubChem CID number | 5354495 | 23952 PubChem SID number | 24852106 | 24855799 SMILES identifier | [Sb] | [Sc] InChI identifier | InChI=1/Sb | InChI=1/Sc RTECS number | CC4025000 | MDL number | MFCD00134030 | MFCD00016323](../image_source/44a85802461ee38c621fe2b7521212fb.png)
| gray antimony | scandium CAS number | 7440-36-0 | 7440-20-2 PubChem CID number | 5354495 | 23952 PubChem SID number | 24852106 | 24855799 SMILES identifier | [Sb] | [Sc] InChI identifier | InChI=1/Sb | InChI=1/Sc RTECS number | CC4025000 | MDL number | MFCD00134030 | MFCD00016323