Input interpretation
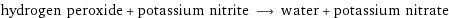
hydrogen peroxide + potassium nitrite ⟶ water + potassium nitrate
Balanced equation

Balance the chemical equation algebraically: + ⟶ + Add stoichiometric coefficients, c_i, to the reactants and products: c_1 + c_2 ⟶ c_3 + c_4 Set the number of atoms in the reactants equal to the number of atoms in the products for H, O, K and N: H: | 2 c_1 = 2 c_3 O: | 2 c_1 + 2 c_2 = c_3 + 3 c_4 K: | c_2 = c_4 N: | c_2 = c_4 Since the coefficients are relative quantities and underdetermined, choose a coefficient to set arbitrarily. To keep the coefficients small, the arbitrary value is ordinarily one. For instance, set c_1 = 1 and solve the system of equations for the remaining coefficients: c_1 = 1 c_2 = 1 c_3 = 1 c_4 = 1 Substitute the coefficients into the chemical reaction to obtain the balanced equation: Answer: | | + ⟶ +
Structures
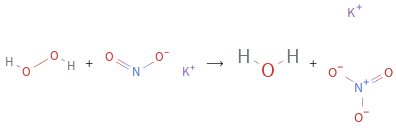
+ ⟶ +
Names
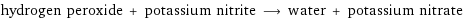
hydrogen peroxide + potassium nitrite ⟶ water + potassium nitrate
Reaction thermodynamics
Gibbs free energy
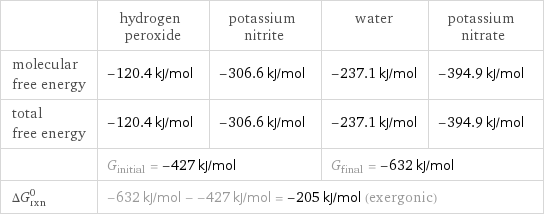
| hydrogen peroxide | potassium nitrite | water | potassium nitrate molecular free energy | -120.4 kJ/mol | -306.6 kJ/mol | -237.1 kJ/mol | -394.9 kJ/mol total free energy | -120.4 kJ/mol | -306.6 kJ/mol | -237.1 kJ/mol | -394.9 kJ/mol | G_initial = -427 kJ/mol | | G_final = -632 kJ/mol | ΔG_rxn^0 | -632 kJ/mol - -427 kJ/mol = -205 kJ/mol (exergonic) | | |
Chemical names and formulas
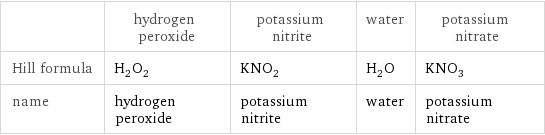
| hydrogen peroxide | potassium nitrite | water | potassium nitrate Hill formula | H_2O_2 | KNO_2 | H_2O | KNO_3 name | hydrogen peroxide | potassium nitrite | water | potassium nitrate
Substance properties
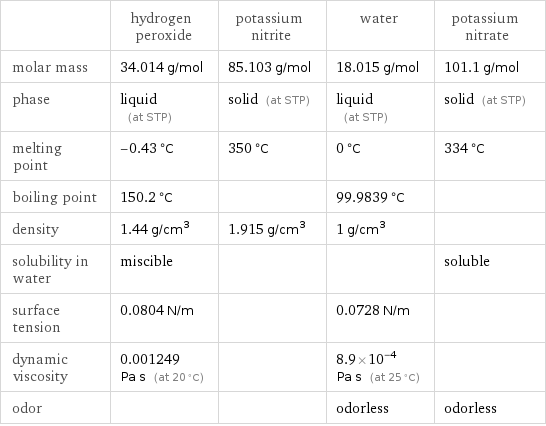
| hydrogen peroxide | potassium nitrite | water | potassium nitrate molar mass | 34.014 g/mol | 85.103 g/mol | 18.015 g/mol | 101.1 g/mol phase | liquid (at STP) | solid (at STP) | liquid (at STP) | solid (at STP) melting point | -0.43 °C | 350 °C | 0 °C | 334 °C boiling point | 150.2 °C | | 99.9839 °C | density | 1.44 g/cm^3 | 1.915 g/cm^3 | 1 g/cm^3 | solubility in water | miscible | | | soluble surface tension | 0.0804 N/m | | 0.0728 N/m | dynamic viscosity | 0.001249 Pa s (at 20 °C) | | 8.9×10^-4 Pa s (at 25 °C) | odor | | | odorless | odorless
Units
