Input interpretation
sodium hydroxide | lithium hydroxide
Chemical names and formulas
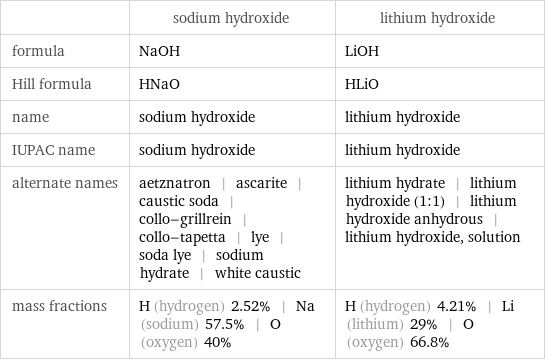
| sodium hydroxide | lithium hydroxide formula | NaOH | LiOH Hill formula | HNaO | HLiO name | sodium hydroxide | lithium hydroxide IUPAC name | sodium hydroxide | lithium hydroxide alternate names | aetznatron | ascarite | caustic soda | collo-grillrein | collo-tapetta | lye | soda lye | sodium hydrate | white caustic | lithium hydrate | lithium hydroxide (1:1) | lithium hydroxide anhydrous | lithium hydroxide, solution mass fractions | H (hydrogen) 2.52% | Na (sodium) 57.5% | O (oxygen) 40% | H (hydrogen) 4.21% | Li (lithium) 29% | O (oxygen) 66.8%
Structure diagrams
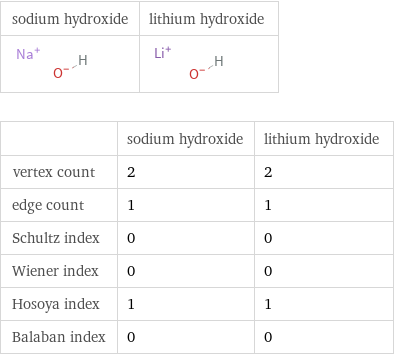
| sodium hydroxide | lithium hydroxide vertex count | 2 | 2 edge count | 1 | 1 Schultz index | 0 | 0 Wiener index | 0 | 0 Hosoya index | 1 | 1 Balaban index | 0 | 0
Basic properties
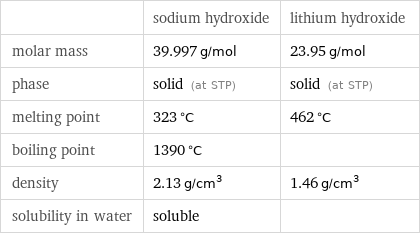
| sodium hydroxide | lithium hydroxide molar mass | 39.997 g/mol | 23.95 g/mol phase | solid (at STP) | solid (at STP) melting point | 323 °C | 462 °C boiling point | 1390 °C | density | 2.13 g/cm^3 | 1.46 g/cm^3 solubility in water | soluble |
Units

Thermodynamic properties
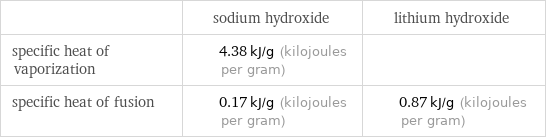
| sodium hydroxide | lithium hydroxide specific heat of vaporization | 4.38 kJ/g (kilojoules per gram) | specific heat of fusion | 0.17 kJ/g (kilojoules per gram) | 0.87 kJ/g (kilojoules per gram)
Solid properties (at STP)
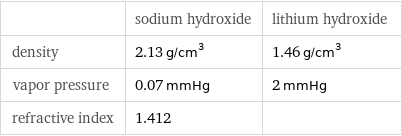
| sodium hydroxide | lithium hydroxide density | 2.13 g/cm^3 | 1.46 g/cm^3 vapor pressure | 0.07 mmHg | 2 mmHg refractive index | 1.412 |
Units
