Input interpretation
lithium (chemical element) | hydrogen (chemical element) | cobalt (chemical element)
Periodic table location
Periodic table location
Images

Images
Basic elemental properties
![| lithium | hydrogen | cobalt atomic symbol | Li | H | Co atomic number | 3 | 1 | 27 short electronic configuration | [He]2s^1 | 1s^1 | [Ar]4s^23d^7 Aufbau diagram | 2s | 1s | 3d 4s block | s | s | d group | 1 | 1 | 9 period | 2 | 1 | 4 atomic mass | 6.94 u | 1.008 u | 58.933194 u half-life | (stable) | (stable) | (stable)](../image_source/3a693bef66c5f7b4819f1725fdbc7632.png)
| lithium | hydrogen | cobalt atomic symbol | Li | H | Co atomic number | 3 | 1 | 27 short electronic configuration | [He]2s^1 | 1s^1 | [Ar]4s^23d^7 Aufbau diagram | 2s | 1s | 3d 4s block | s | s | d group | 1 | 1 | 9 period | 2 | 1 | 4 atomic mass | 6.94 u | 1.008 u | 58.933194 u half-life | (stable) | (stable) | (stable)
Thermodynamic properties
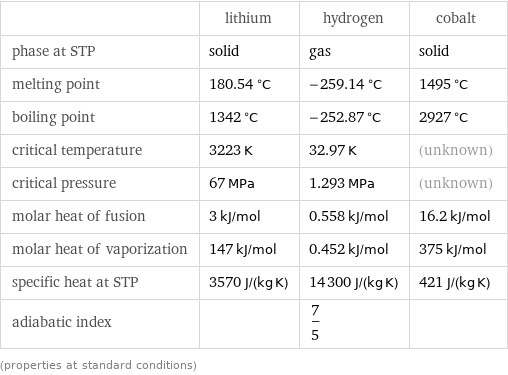
| lithium | hydrogen | cobalt phase at STP | solid | gas | solid melting point | 180.54 °C | -259.14 °C | 1495 °C boiling point | 1342 °C | -252.87 °C | 2927 °C critical temperature | 3223 K | 32.97 K | (unknown) critical pressure | 67 MPa | 1.293 MPa | (unknown) molar heat of fusion | 3 kJ/mol | 0.558 kJ/mol | 16.2 kJ/mol molar heat of vaporization | 147 kJ/mol | 0.452 kJ/mol | 375 kJ/mol specific heat at STP | 3570 J/(kg K) | 14300 J/(kg K) | 421 J/(kg K) adiabatic index | | 7/5 | (properties at standard conditions)
Material properties
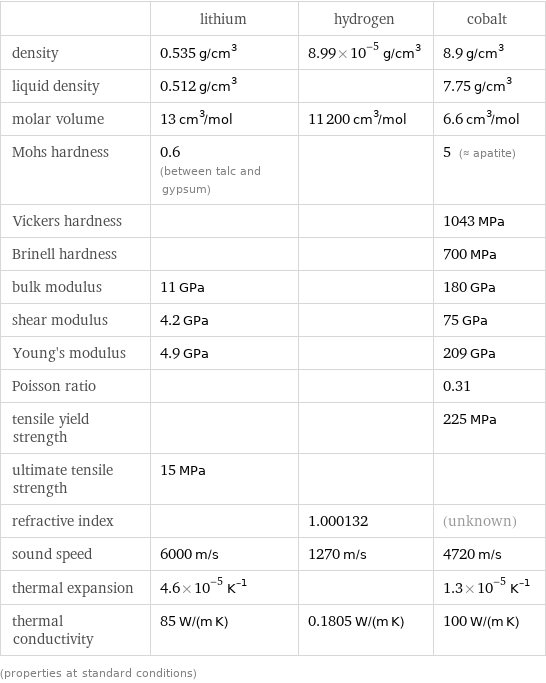
| lithium | hydrogen | cobalt density | 0.535 g/cm^3 | 8.99×10^-5 g/cm^3 | 8.9 g/cm^3 liquid density | 0.512 g/cm^3 | | 7.75 g/cm^3 molar volume | 13 cm^3/mol | 11200 cm^3/mol | 6.6 cm^3/mol Mohs hardness | 0.6 (between talc and gypsum) | | 5 (≈ apatite) Vickers hardness | | | 1043 MPa Brinell hardness | | | 700 MPa bulk modulus | 11 GPa | | 180 GPa shear modulus | 4.2 GPa | | 75 GPa Young's modulus | 4.9 GPa | | 209 GPa Poisson ratio | | | 0.31 tensile yield strength | | | 225 MPa ultimate tensile strength | 15 MPa | | refractive index | | 1.000132 | (unknown) sound speed | 6000 m/s | 1270 m/s | 4720 m/s thermal expansion | 4.6×10^-5 K^(-1) | | 1.3×10^-5 K^(-1) thermal conductivity | 85 W/(m K) | 0.1805 W/(m K) | 100 W/(m K) (properties at standard conditions)
Electromagnetic properties
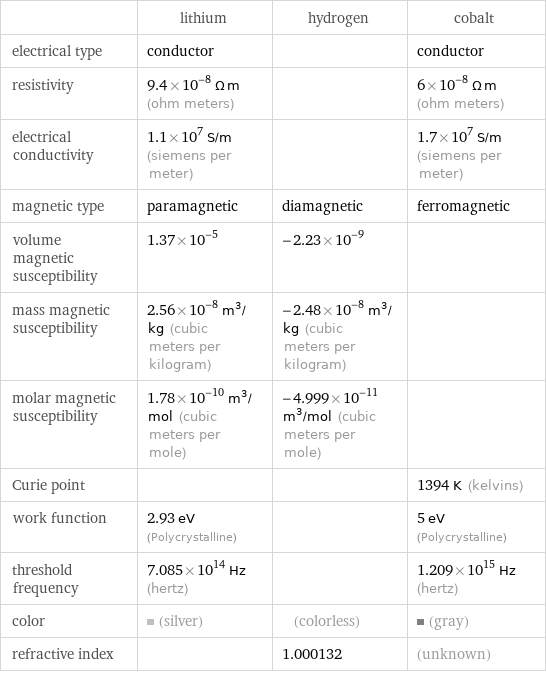
| lithium | hydrogen | cobalt electrical type | conductor | | conductor resistivity | 9.4×10^-8 Ω m (ohm meters) | | 6×10^-8 Ω m (ohm meters) electrical conductivity | 1.1×10^7 S/m (siemens per meter) | | 1.7×10^7 S/m (siemens per meter) magnetic type | paramagnetic | diamagnetic | ferromagnetic volume magnetic susceptibility | 1.37×10^-5 | -2.23×10^-9 | mass magnetic susceptibility | 2.56×10^-8 m^3/kg (cubic meters per kilogram) | -2.48×10^-8 m^3/kg (cubic meters per kilogram) | molar magnetic susceptibility | 1.78×10^-10 m^3/mol (cubic meters per mole) | -4.999×10^-11 m^3/mol (cubic meters per mole) | Curie point | | | 1394 K (kelvins) work function | 2.93 eV (Polycrystalline) | | 5 eV (Polycrystalline) threshold frequency | 7.085×10^14 Hz (hertz) | | 1.209×10^15 Hz (hertz) color | (silver) | (colorless) | (gray) refractive index | | 1.000132 | (unknown)
Reactivity
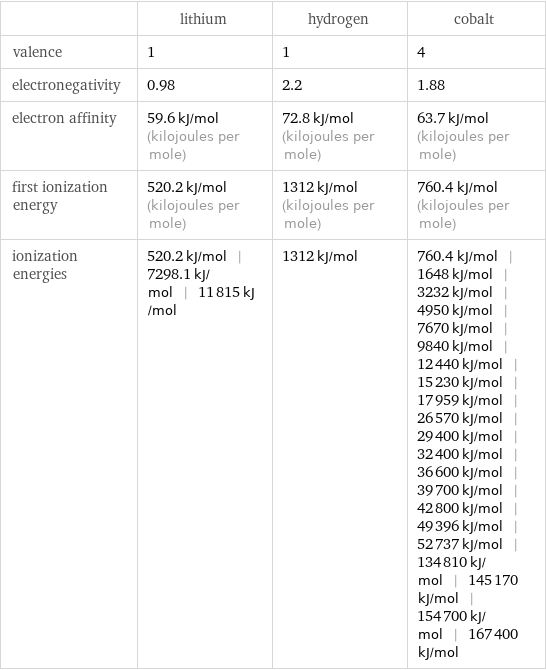
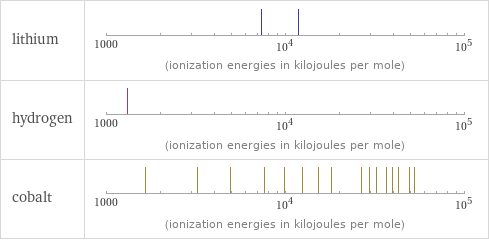
| lithium | hydrogen | cobalt valence | 1 | 1 | 4 electronegativity | 0.98 | 2.2 | 1.88 electron affinity | 59.6 kJ/mol (kilojoules per mole) | 72.8 kJ/mol (kilojoules per mole) | 63.7 kJ/mol (kilojoules per mole) first ionization energy | 520.2 kJ/mol (kilojoules per mole) | 1312 kJ/mol (kilojoules per mole) | 760.4 kJ/mol (kilojoules per mole) ionization energies | 520.2 kJ/mol | 7298.1 kJ/mol | 11815 kJ/mol | 1312 kJ/mol | 760.4 kJ/mol | 1648 kJ/mol | 3232 kJ/mol | 4950 kJ/mol | 7670 kJ/mol | 9840 kJ/mol | 12440 kJ/mol | 15230 kJ/mol | 17959 kJ/mol | 26570 kJ/mol | 29400 kJ/mol | 32400 kJ/mol | 36600 kJ/mol | 39700 kJ/mol | 42800 kJ/mol | 49396 kJ/mol | 52737 kJ/mol | 134810 kJ/mol | 145170 kJ/mol | 154700 kJ/mol | 167400 kJ/mol
Reactivity
Atomic properties
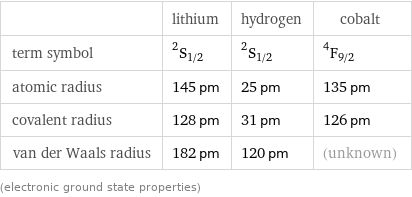
| lithium | hydrogen | cobalt term symbol | ^2S_(1/2) | ^2S_(1/2) | ^4F_(9/2) atomic radius | 145 pm | 25 pm | 135 pm covalent radius | 128 pm | 31 pm | 126 pm van der Waals radius | 182 pm | 120 pm | (unknown) (electronic ground state properties)
Abundances
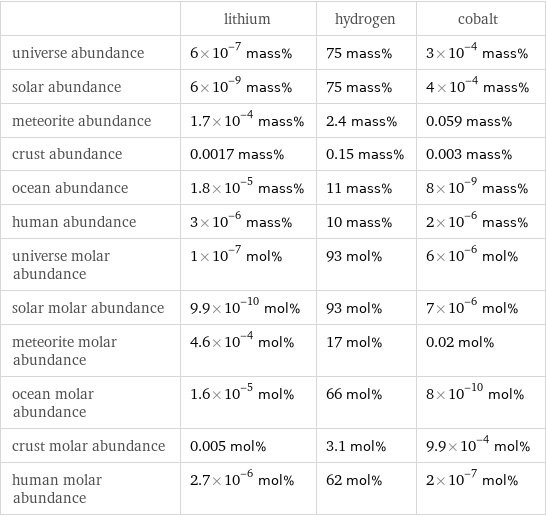
| lithium | hydrogen | cobalt universe abundance | 6×10^-7 mass% | 75 mass% | 3×10^-4 mass% solar abundance | 6×10^-9 mass% | 75 mass% | 4×10^-4 mass% meteorite abundance | 1.7×10^-4 mass% | 2.4 mass% | 0.059 mass% crust abundance | 0.0017 mass% | 0.15 mass% | 0.003 mass% ocean abundance | 1.8×10^-5 mass% | 11 mass% | 8×10^-9 mass% human abundance | 3×10^-6 mass% | 10 mass% | 2×10^-6 mass% universe molar abundance | 1×10^-7 mol% | 93 mol% | 6×10^-6 mol% solar molar abundance | 9.9×10^-10 mol% | 93 mol% | 7×10^-6 mol% meteorite molar abundance | 4.6×10^-4 mol% | 17 mol% | 0.02 mol% ocean molar abundance | 1.6×10^-5 mol% | 66 mol% | 8×10^-10 mol% crust molar abundance | 0.005 mol% | 3.1 mol% | 9.9×10^-4 mol% human molar abundance | 2.7×10^-6 mol% | 62 mol% | 2×10^-7 mol%
Nuclear properties

| lithium | hydrogen | cobalt half-life | (stable) | (stable) | (stable) stable isotopes | Li-7 (92.41%) | Li-6 (7.59%) | H-1 (99.985%) | H-2 (1.5×10^-4) | Co-59 (100%) nuclear spin | Li-4: 2^- | Li-5: 3/2^- | Li-6: 1^+ | Li-7: 3/2^- | Li-8: 2^+ | Li-9: 3/2^- | Li-11: 3/2^- | H-1: 1/2^+ | H-2: 1^+ | H-3: 1/2^+ | H-4: 2^- | H-5: 1/2^+ | H-6: 2^- | H-7: 1/2^+ | Co-47: 7/2^- | Co-48: 6^+ | Co-49: 7/2^- | Co-50: 6^+ | Co-51: 7/2^- | Co-52: 6^+ | Co-53: 7/2^- | Co-54: 0^+ | Co-55: 7/2^- | Co-56: 4^+ | Co-57: 7/2^- | Co-58: 2^+ | Co-59: 7/2^- | Co-60: 5^+ | Co-61: 7/2^- | Co-62: 2^+ | Co-63: 7/2^- | Co-64: 1^+ | Co-65: 7/2^- | Co-66: 3^+ | Co-67: 7/2^- | Co-68: 7^- | Co-69: 7/2^- | Co-70: 6^- | Co-71: 7/2^- | Co-73: 7/2^- | Co-74: 0^+ | Co-75: 7/2^- unstable isotopes | Li-8 (839.9 ms) | Li-9 (178.3 ms) | Li-11 (8.59 ms) | Li-12 (10 ns) | Li-10 (2 zs) | Li-5 (0.3 zs) | Li-4 (0.0757 zs) | H-3 (12.32 yr) | H-6 (0.29 zs) | H-4 (0.139 zs) | H-5 (0.08 zs) | H-7 (0.023 zs) | Co-60 (5.2749 yr) | Co-57 (271.74 days) | Co-56 (77.23 days) | Co-58 (70.86 days) | Co-55 (17.531 h) | Co-61 (99 min) | Co-62 (90 s) | Co-63 (27.4 s) | Co-65 (1.2 s) | Co-67 (425 ms) | Co-64 (300 ms) | Co-53 (240 ms) | Co-69 (220 ms) | Co-68 (200 ms) | Co-54 (193.28 ms) | Co-66 (180 ms) | Co-70 (119 ms) | Co-52 (115 ms) | Co-71 (79 ms) | Co-72 (62 ms) | Co-50 (44 ms) | Co-73 (41 ms) | Co-51 (200 ns) | Co-75 (150 ns) | Co-74 (150 ns) | Co-49 (35 ns) neutron cross-section | 71 b | 0.332 b | 37.2 b neutron mass absorption | | 0.011 m^2/kg | 0.021 m^2/kg
Identifiers
| lithium | hydrogen | cobalt CAS number | 7439-93-2 | 1333-74-0 | 7440-48-4 PubChem CID number | 3028194 | 783 | 104730