Input interpretation
copper(II) nitrate | stannous chloride dihydrate
Chemical names and formulas
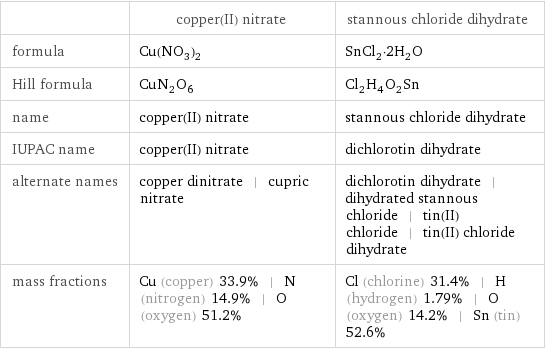
| copper(II) nitrate | stannous chloride dihydrate formula | Cu(NO_3)_2 | SnCl_2·2H_2O Hill formula | CuN_2O_6 | Cl_2H_4O_2Sn name | copper(II) nitrate | stannous chloride dihydrate IUPAC name | copper(II) nitrate | dichlorotin dihydrate alternate names | copper dinitrate | cupric nitrate | dichlorotin dihydrate | dihydrated stannous chloride | tin(II) chloride | tin(II) chloride dihydrate mass fractions | Cu (copper) 33.9% | N (nitrogen) 14.9% | O (oxygen) 51.2% | Cl (chlorine) 31.4% | H (hydrogen) 1.79% | O (oxygen) 14.2% | Sn (tin) 52.6%
Structure diagrams
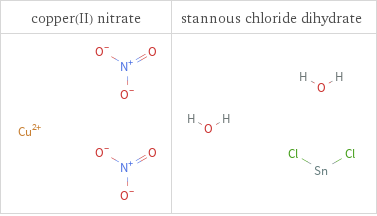
Structure diagrams
Basic properties
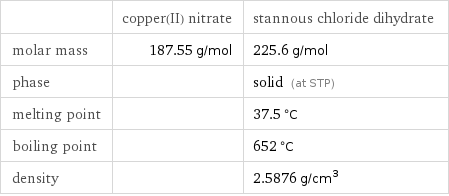
| copper(II) nitrate | stannous chloride dihydrate molar mass | 187.55 g/mol | 225.6 g/mol phase | | solid (at STP) melting point | | 37.5 °C boiling point | | 652 °C density | | 2.5876 g/cm^3
Units
