Input interpretation
inorganic salts | molar heat of fusion
Summary

median | 23.3 kJ/mol highest | 58.6 kJ/mol (ammonium nitrate) lowest | 0.15 kJ/mol (nickel(II) nitrate) distribution | | (based on 19 values; 1 unavailable)
Entity with missing value
sodium hyposulfite
Distribution plots
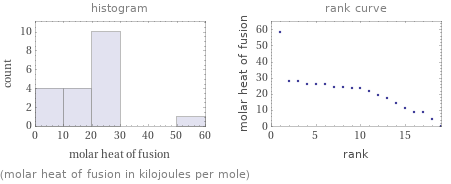
(molar heat of fusion in kilojoules per mole)
Molar heat of fusion rankings

1 | nickel(II) nitrate | 0.15 kJ/mol 2 | rubidium nitrate | 4.6 kJ/mol 3 | lithium sulfate | 9 kJ/mol 4 | beryllium chloride | 8.66 kJ/mol 5 | ammonium bisulfate | 10.92 kJ/mol 6 | lithium iodide | 14.6 kJ/mol 7 | lithium bromide | 17.66 kJ/mol 8 | sodium sulfide | 19 kJ/mol 9 | cesium fluoride | 21.7 kJ/mol 10 | rubidium bromide | 23.3 kJ/mol 11 | sodium sulfate | 23.85 kJ/mol 12 | rubidium chloride | 24.4 kJ/mol 13 | potassium iodide | 24 kJ/mol 14 | rubidium fluoride | 25.8 kJ/mol 15 | sodium bromide | 26.23 kJ/mol 16 | potassium chloride | 26.28 kJ/mol 17 | sodium bromate | 28.11 kJ/mol 18 | sodium chloride | 28.16 kJ/mol 19 | ammonium nitrate | 58.6 kJ/mol (based on 19 values; 1 unavailable)
Unit conversions for median molar heat of fusion 23.3 kJ/mol
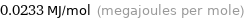
0.0233 MJ/mol (megajoules per mole)
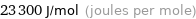
23300 J/mol (joules per mole)

5.57 kcal/mol (thermochemical kilocalories per mole)
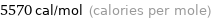
5570 cal/mol (calories per mole)

0.241 eV (molar electronvolts)