Input interpretation
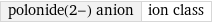
polonide(2-) anion | ion class
Result
anions | group 16 ions | p block ions
Ionic radii
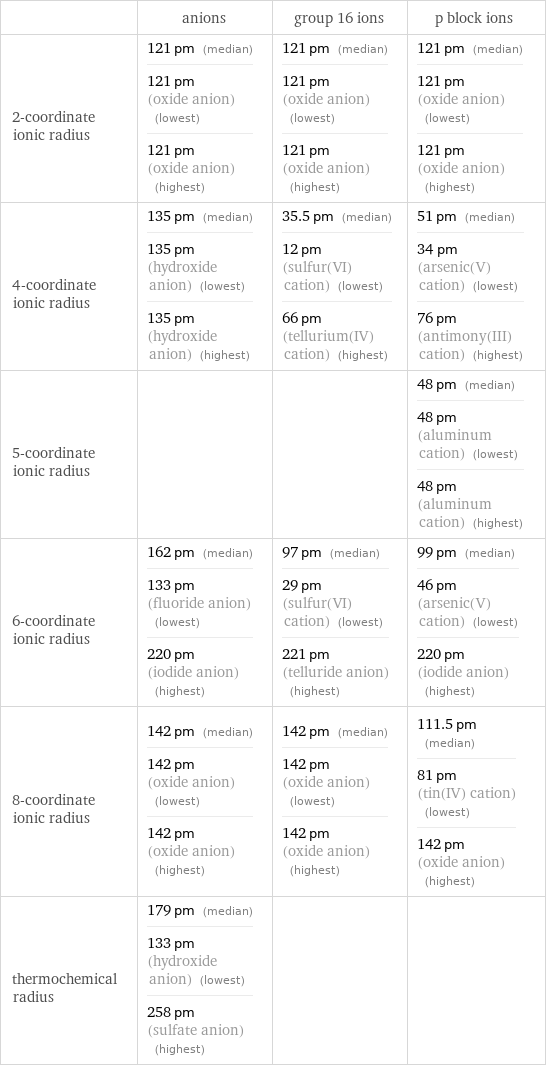
| anions | group 16 ions | p block ions 2-coordinate ionic radius | 121 pm (median) 121 pm (oxide anion) (lowest) 121 pm (oxide anion) (highest) | 121 pm (median) 121 pm (oxide anion) (lowest) 121 pm (oxide anion) (highest) | 121 pm (median) 121 pm (oxide anion) (lowest) 121 pm (oxide anion) (highest) 4-coordinate ionic radius | 135 pm (median) 135 pm (hydroxide anion) (lowest) 135 pm (hydroxide anion) (highest) | 35.5 pm (median) 12 pm (sulfur(VI) cation) (lowest) 66 pm (tellurium(IV) cation) (highest) | 51 pm (median) 34 pm (arsenic(V) cation) (lowest) 76 pm (antimony(III) cation) (highest) 5-coordinate ionic radius | | | 48 pm (median) 48 pm (aluminum cation) (lowest) 48 pm (aluminum cation) (highest) 6-coordinate ionic radius | 162 pm (median) 133 pm (fluoride anion) (lowest) 220 pm (iodide anion) (highest) | 97 pm (median) 29 pm (sulfur(VI) cation) (lowest) 221 pm (telluride anion) (highest) | 99 pm (median) 46 pm (arsenic(V) cation) (lowest) 220 pm (iodide anion) (highest) 8-coordinate ionic radius | 142 pm (median) 142 pm (oxide anion) (lowest) 142 pm (oxide anion) (highest) | 142 pm (median) 142 pm (oxide anion) (lowest) 142 pm (oxide anion) (highest) | 111.5 pm (median) 81 pm (tin(IV) cation) (lowest) 142 pm (oxide anion) (highest) thermochemical radius | 179 pm (median) 133 pm (hydroxide anion) (lowest) 258 pm (sulfate anion) (highest) | |