Input interpretation
uranium dioxide
Chemical names and formulas

formula | UO_2 Hill formula | O_2U name | uranium dioxide IUPAC name | dioxouranium alternate names | uranium(4) oxide, red | uranium(IV) oxide | uranium oxide | uranium oxide (1:2) mass fractions | O (oxygen) 11.8% | U (uranium) 88.2%
Structure diagram
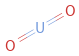
Structure diagram
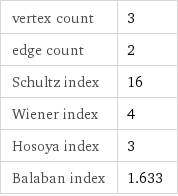
vertex count | 3 edge count | 2 Schultz index | 16 Wiener index | 4 Hosoya index | 3 Balaban index | 1.633
Basic properties
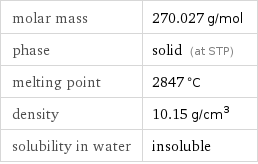
molar mass | 270.027 g/mol phase | solid (at STP) melting point | 2847 °C density | 10.15 g/cm^3 solubility in water | insoluble
Units

Solid properties (at STP)

density | 10.15 g/cm^3
Units

Thermodynamic properties
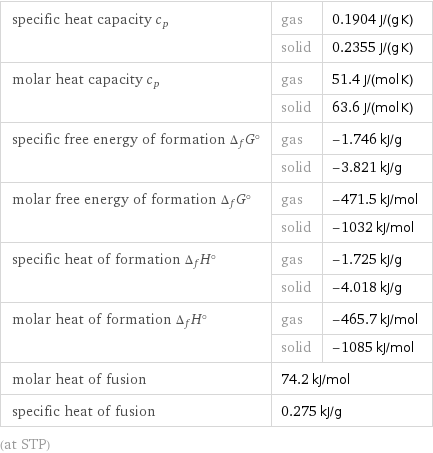
specific heat capacity c_p | gas | 0.1904 J/(g K) | solid | 0.2355 J/(g K) molar heat capacity c_p | gas | 51.4 J/(mol K) | solid | 63.6 J/(mol K) specific free energy of formation Δ_fG° | gas | -1.746 kJ/g | solid | -3.821 kJ/g molar free energy of formation Δ_fG° | gas | -471.5 kJ/mol | solid | -1032 kJ/mol specific heat of formation Δ_fH° | gas | -1.725 kJ/g | solid | -4.018 kJ/g molar heat of formation Δ_fH° | gas | -465.7 kJ/mol | solid | -1085 kJ/mol molar heat of fusion | 74.2 kJ/mol | specific heat of fusion | 0.275 kJ/g | (at STP)
Chemical identifiers
![CAS number | 1344-57-6 PubChem CID number | 10916 SMILES identifier | O=[U]=O EU number | 215-700-3 Gmelin number | 9351](../image_source/00a6bd3dc09edddd4aa9ebb563b60317.png)
CAS number | 1344-57-6 PubChem CID number | 10916 SMILES identifier | O=[U]=O EU number | 215-700-3 Gmelin number | 9351